Quality, Safety and Value (QSVI)
Local Infiltration Anesthesia with Liposomal Bupivacaine Decreases Postoperative Narcotic Consumption in AIS
Scottish Rite for Children, Dallas, TX
Correspondence: Amy L. McIntosh, MD, Scottish Rite for Children, 2222 Welborn St., Dallas, TX 75219. E-mail: [email protected]
Received: February 11, 2022; Accepted: March 14, 2022; Published: May 1, 2022
DOI: 10.55275/JPOSNA-2022-0024
Volume 4, Number 2, May 2022
Abstract:
Background: Our previous standardized postoperative pain management plan for AIS patients undergoing PSF included the placement of an epidural (EPI) catheter by the spine surgeon prior to fascial closure. The epidural was used for 24 hours in conjunction with a multi-modal postoperative pain protocol.
Local Problem: The 2020 COVID-19 pandemic highlighted the need for our surgical AIS patients and their families to have private rooms/bathrooms. Epidural catheters require arterial line blood pressure monitoring to ensure safety. Unfortunately, the arterial lines could not be utilized in the private rooms due to lack of appropriate monitors, therefore we transitioned away from epidurals.
Liposomal bupivacaine (LB) is an encapsulated local anesthetic that has a duration of action up to 72 hours.
Specific Aims: This quality initiative (QI) focused on the enhancement of the standardized postoperative care pathway for AIS patients undergoing PSF. The aims of the initiative were to 1) minimize narcotic usage, and 2) allow for earlier mobilization.
Intervention: The (2020) LB cohort had a mixture of LB, bupivacaine HCL and normal saline injected into the fascial and subcutaneous layers of the incision prior to fascial closure. The (2019) control cohort had an epidural (EPI) catheter placed prior to fascial closure. The epidural infusion consisted of ropivacaine 0.1% (0.1–0.2 ml/kg/hr) The cohorts received the same pain protocol (intravenous opioid with transition to oral opioid, as well as dexmedetomidine, acetaminophen, ketorolac, and diazepam).
Measures and Analysis: Two cohorts of prospective AIS patients that underwent PSF were retrospectively reviewed. They were compared regarding age, BMI, average preoperative major Cobb angle, average preoperative kyphosis, VAS pain scores, and narcotic consumption. Time to ambulation and length of stay (LOS) were also evaluated.
Results: 159 AIS patients were reviewed. There were no preoperative differences between the 2020 LB cohort (n=101) and the 2019 EPI cohort (n=58) when comparing age (14.3 vs. 15.0 years), average BMI (21.7 vs. 20.8 kg/m2), preoperative major Cobb (60.7 degrees vs. 60.3 degrees), or average preoperative kyphosis (25.7 degrees vs. 27.1 degrees). The LB cohort consumed similar morphine equivalents during the initial 24 hours (37.8 mg vs. 37.3 mg) but less at 24–48 hours (37.2 mg vs. 41.0 mg) (p=0.031), and 48-72 hours (23.3 mg vs. 29.6 mg) (p=0.015). The LB cohort ambulated 6.8 hours sooner (17.1 vs. 23.9 hrs) (p=.0001).
Summary: LB associated with less consumption of postoperative morphine equivalents and earlier ambulation.
Introduction
Standardized postoperative care pathways reduce practice variability and allow for meaningful assessment of outcomes in specific patient populations. Over the last 5 years, multiple standardized postoperative care pathways for patients undergoing posterior spinal fusion (PSF) for the treatment of adolescent idiopathic scoliosis (AIS) have been published.1–4 A key feature of these AIS care pathways is a multimodal approach to pain control. Several non-opioid analgesic medications, including gabapentinoids, acetaminophen (paracetamol), nonsteroidal anti-inflammatory drugs (NSAIDs), and muscle relaxants (Valium [diazepam]), are given in combination with opioid medications. This, in turn, allows for earlier mobilization and ambulation as well as decreased lengths of stay.
Multimodal analgesic regimens, which include non-opioid regional anesthetics, are being implemented by hospitals to manage postsurgical pain and improve recovery and to also minimize in-hospital postsurgical opioid consumption.5
Liposomal bupivacaine (LB) is a long-acting single-dose infiltration multivesicular liposome formulation of the local anesthetic bupivacaine that provides prolonged bupivacaine release over several days that has recently gained FDA approval in the pediatric population.6 A recent retrospective analysis of the de-identified Premier Healthcare Database for patients aged 1–17 years who underwent inpatient primary spine surgery, including discectomy, lumbosacral fusion, other fusion, laminectomy, or other spine surgery, demonstrated that the liposomal bupivacaine (LB) cohort (n=373) had significantly reduced in-hospital postsurgical opioid consumption, LOS, and hospital costs compared with the non-LB cohort (n=9816).
Local Problem
The previous standardized postoperative care pathway for AIS patients undergoing PSF at our institution included the placement of an epidural (EPI) catheter by the spine surgeon prior to fascial closure. The continuous epidural infusion consisted of ropivacaine 0.1% at a range of 0.1-0.2 ml/kg/hr. The epidural was used for the first 24 hours postoperatively in conjunction with a multi-modal postoperative pain protocol (intravenous opioid with transition to oral opioid as well as IV dexmedtomidine, acetaminophen, ketorolac, and diazepam). Our hospital does not have an Intensive Care Unit (ICU), and all surgical spine patients were managed on the orthopaedic floor in a separate Intermediate Care Area (ICA). The patients were hemodynamically monitored (arterial line blood pressure monitoring, foley catheters, heart rate, respiratory rate, and pulse oximetry). The ICA has five rooms and a shared common bathroom for patients and their families.
However, the 2020 COVID-19 pandemic forced us to quickly change our standardized postoperative care pathway for AIS patients undergoing PSF. This was done to allow patients and their family members to have private rooms with individual private bathrooms. This also allowed for appropriate social distancing when outside of their rooms. Epidurals and IV dexmedtomidine require arterial line blood pressure monitoring to ensure safety. Unfortunately, arterial lines could not be monitored in the private rooms due to lack of appropriate monitors; therefore, we transitioned away from epidurals to liposomal bupivacaine (LB).
Specific Aims
This quality initiative (QI) focused on the enhancement of the patient-centered interdisciplinary standardized postoperative care pathway for AIS patients undergoing PSF. The aims of the initiative were to 1) minimize narcotic usage, and 2) allow for earlier mobilization.
Intervention
Prior to wound closure, the LB cohort (all surgeries were performed in the year 2020) had local infiltration anesthesia (LIA) performed by the surgeon prior to fascial closure. The mixture of liposomal bupivacaine, bupivacaine HCL and normal saline was injected into the fascial and subcutaneous layers of the incision. For patients less than 60 kg, 4 mg/kg of LB was mixed with 2 mg/kg 0.25% bupivacaine. Normal saline was added to create up to a 1:14 dilution by volume depending on incision length. Patients greater than 60 kg received 266 mg of LB, 125 mg of 0.25% bupivacaine and additional normal saline up to a 1:14 dilution by volume for adequate incision coverage. Ideally, 30 ml of solution is infiltrated every 2.5 cm of incision. Two-thirds of the total solution was injected into the fascial layer and one-third into the subcutaneous layer.
The control cohort had an epidural (EPI) catheter placed by the surgeon prior to fascial closure. The continuous epidural infusion consisted of ropivacaine 0.1% at a range of 0.1-0.2 ml/kg/hr. All patients in the EPI cohort had surgery performed in the year 2019.
All patients in both cohorts received one IV dose (4 mg) of dexamethasone intraoperatively for prophylaxis to help control postoperative nausea and vomiting. No postoperative doses of dexamethasone were given.7 Our standard postoperative posterior spinal fusion pathway for patients with AIS included intravenous opioid (hydromorphone) as needed (no basal rate) with transition to an oral opioid (oxycodone or hydrocodone) within the first 24 hours postoperatively. Intravenous ketorolac was given every 6 hours for 24 hours with transition to oral ibuprofen; intravenous acetaminophen was given every 6 hours for 24 hours with transition to oral acetaminophen. Oral diazepam was available as needed for muscle spasms. For the first night after surgery, low dose dexmedetomidine was used as an adjunctive analgesic and sedative.
Measures and Analysis
A multi-disciplinary team (surgeon, anesthesia, EPIC analyst, director of quality) was assembled, and surgical data collection capabilities were built into the EMR (EPIC). Two cohorts of AIS patients that underwent PSF were extracted and compared on a bimonthly basis. The team reviewed the data, and changes were made to extraction to ensure high quality/accurate data.
We reviewed the records of patients 10 to 18 years of age who had undergone posterior spinal fusion for the treatment of AIS at a single academic institution. Variables that were collected included the LOS (starting from the time of admission to the postop anesthesia care unit (PACU) until hospital discharge), the total opioid dosage administered (converted to oral MME with use of the conversion table of the Centers for Disease Control and Prevention [CDC], and average visual analog scale (VAS) pain scores during the following time points (0-24 hrs and 25-48 hrs). Mobility was measured as time to first ambulation whether in the room or out into the hallway.
Statistical analysis was performed to compare the control and intervention groups using a two-sample t-test. Significance was defined as p <0.05. IRB approval was not necessary for this QI.
Results
One hundred fifty-nine AIS patients underwent PSF. There were no preoperative statistically significant differences between the 2020 LB cohort (n=101) and the 2019 EPI cohort (n=58) when comparing age (14.3 vs.15.0 years), average BMI (21.7 vs. 20.8 kg/m2), preoperative major Cobb (60.7 degrees vs. 60.3 degrees), or average preoperative kyphosis (25.7 degrees vs. 27.1 degrees). Average VAS pain scores at 0-24 hours, and 25-48 hours did NOT differ between the 2 cohorts (1.8 vs. 1.9, and 1.8 vs. 2.0) (p=0.421, 0.052). The LB cohort consumed similar morphine equivalents during the initial 24 hours (37.8 mg vs. 37.3 mg), but less at 24-48 hours (37.2 mg vs. 41.0 mg) (p=0.031), and 48-72 hours (23.3 mg vs. 29.6 mg) (p=0.015).
The LB cohort ambulated 6.8 hours sooner (17.1 vs. 23.9 hrs) (p=0001), and the LB cohort’s length of stay (LOS) was 5 hours less.
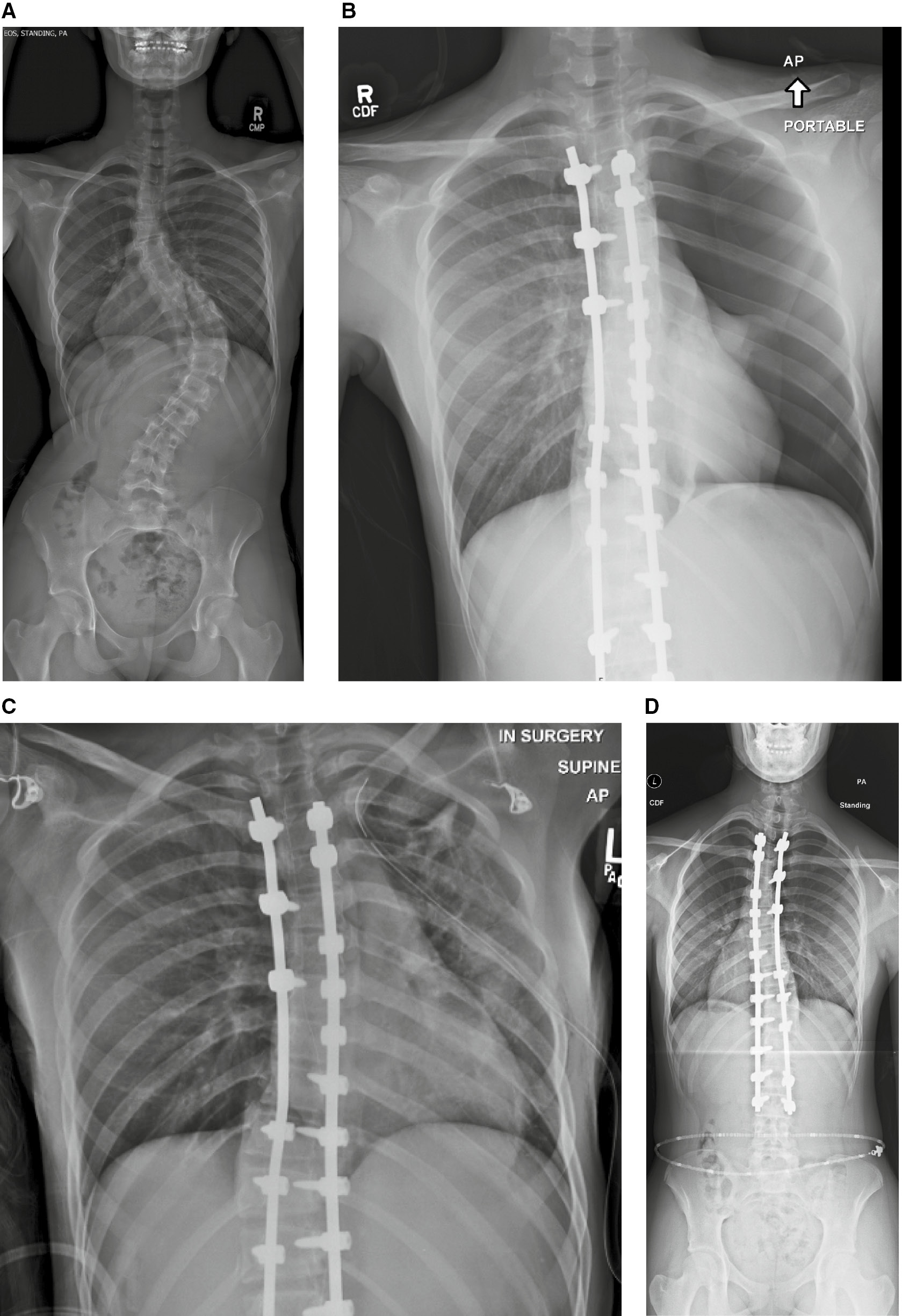
Three patients (3/101: 2.97%) in the LB cohort developed an iatrogenic pneumothorax of the convex chest wall (Figure 1). Root Cause Analysis (RCA) determine the cause to be due to inadvertent needle penetration during the infiltration of the fascial musculature with LB. All three patients were diagnosed while in the hospital due to shortness of breath (SOB) and decreasing oxygen saturation on pulse oximetry. They were all treated with chest tube or pigtail catheter placement. We changed the needle from a 1.5-inch 20-gauge hypodermic needle to a 1-inch 20-gauge hypodermic needle and following that change we have not had any further inadvertent needle penetrations causing a pneumothorax. No pneumothoraxes occurred in the epidural group.
Figure 1. (A-D) 15-year-old female with AIS who had iatrogenic pneumothorax of the convex chest wall that occurred during LB infiltration. This was treated with insertion of pig-tail catheter by interventional radiology.
Discussion
A recent study by Chughtai et al. demonstrated similar decreased narcotic consumption in pediatric patients that had undergone posterior spinal deformity correction surgery and underwent LIA with LB.8 In their study, both the LB and non-LB cohorts had two epidural catheters placed intraoperatively and received epidural analgesia for a maximum of 48 hours postoperatively. In contrast, this study compares a LB cohort to an epidural (EPI) cohort (no LB). Interestingly, the VAS pain scores and narcotic consumption were equivalent between the two groups during the first 24 hours postoperatively (VAS pain scores: 1.9 vs. 1.8) (p=0.421) (morphine equivalents: 37.8 vs. 37.3 mg) (p=0.781). Therefore, in this AIS patient population, LIA with LB was equivalent to a ropivacaine 0.1% epidural in the first 24 hours postoperatively. We no longer place epidurals for postoperative pain management in AIS patients.
The Chughtai study did not report any iatrogenic pneumothoraxes. They reported using a 20-gauge needle but not the length. We recommend using a 1-inch hypodermic 20-gauge needle and having an awareness of the residual chest wall rotation following spinal deformity correct. The needle should be placed into the fascial layer 1-1.5 cm in depth and the needle trajectory should be superficial to the ribs.
In another recent study by Cohen et al., pediatric patients undergoing open or laparoscopic surgery received wound infiltration/ local infiltration anesthesia. They compared the incidence of local anesthetic systemic toxicity among those who received any dose of liposomal bupivacaine and those who received plain bupivacaine.9 They had no cases of local anesthetic systemic toxicity syndrome (LAST) and few patients with any sign or symptom that could potentially be related to local anesthetic toxicity. We also had no cases of LAST or local anesthetic toxicity.
This cohort-controlled comparison highlights that local infiltration anesthesia (LIA) using liposomal bupivacaine (LB) improved our AIS Post-Operative Rapid Recovery Program. The LB cohort was equivalent to the EPI cohort in the first 24 hours postoperatively when comparing VAS pain scores and consumption of morphine equivalents. During the remainder of the in-patient hospitalization, VAS pain scores remained equivalent between the two groups. However, the LB cohort consumed less morphine equivalents than the EPI cohort: 24-48 hours (37.2 mg vs. 41.0 mg) (p=0.031), and 48-72 hours (23.3 mg vs. 29.6 mg) (p=0.015). The LB cohort also demonstrated earlier mobilization/ambulation compared to the EPI cohort (17.1 vs. 23.9 hrs) (p=.0001).
Conclusion
This information has led to a significant change in our institution’s standardized AIS Post-Operative Rapid Recovery Program. We no longer routinely place epidurals for the AIS patient population, arterial lines are removed in the PACU, and these patients go immediately to private rooms on the orthopaedic floor. We continue to refine our AIS Post-Operative Rapid Recovery Program with other quality initiatives. Our goals remain consistent: 1) minimize narcotic usage, 2) allow for earlier mobilization, and 3) decrease length of stay.
Disclaimer
A. L. McIntosh: Consultant for NuVasive. C. McLeod has no conflicts of interest to report.
References
- Yang J SD, Chan P, Villamor GA, et al. High satisfaction in adolescent idiopathic scoliosis patients on enhanced discharge pathway . J Pediatr Orthop. 2020;40(3):e166-e170.
- Oetgen ME, Martin BD, Gordish-Dressman H, et al. Effectiveness and sustainability of a standardized care pathway developed with use of lean processmapping for the treatment of patients undergoing posterior spinal fusion for adolescent idiopathic scoliosis. J Bone Joint Surg Am. 2018;100(21):1864-1870.
- Sanders AE, Andras LM, Sousa T, et al. Accelerated discharge protocol for posterior spinal fusion patients with adolescent idiopathic scoliosis decreases hospital postoperative charges. Spine (Phila Pa 1976). 2017;15(42):92-97.
- Gornitzky AL, Flynn JM, Muhly WT, et al. A rapid recovery pathway for adolescent idiopathic scoliosis that improves pain control and reduces time to inpatient recovery after posterior spinal fusion. Spine Deform. 2016;4(4):288–295.
- Ballock RT, Seif J, Goodwin R, Lin JH, Cirillo J. Clinical and economic outcomes associated with use of liposomal bupivacaine versus standard of care for management of postsurgical pain in pediatric patients undergoing spine surgery. J Health Econ. Outcomes Res 2021;8(1):29.
- Tirotta CF, de Armendi AJ, Horn ND, et al. A multicenter study to evaluate the pharmacokinetics and safety of liposomal bupivacaine for postsurgical analgesia in pediatric patients aged 6 to less than 17 years (PLAY). J Clin Anesthesia 2021;75(3):110503.
- Fletcher ND, Ruska T, Austin TM, et al. Postoperative dexamethasone following posterior spinal fusion for adolescent idiopathic scoliosis. J Bone Joint Surg Am. 2020;102(20):1807-1813.
- Chughtai M, Sultan AA, Hudson B, et al. Liposomal bupivacaine is both safe and effective in controlling postoperative pain after spinal surgery in children: a controlled cohort study. Clin Spine Surg. 2020;33(10):533-538.
- Cohen B, Glosser L, Saab R, et al. Incidence of adverse events attributable to bupivacaine liposome injectable suspension or plain bupivacaine for postoperative pain in pediatric surgical patients: a retrospective matched cohort analysis. Pediatr Anesth. 2019;29(2):169-174.
