Current Concept Review
Regional and Local Anesthesia in Pediatric Orthopaedic Surgery
1Nemours Children’s Health, Orlando, FL; 2University of Central Florida School of Medicine, Orlando, FL; 3Connecticut Children’s, Hartford, CT; 4Uconn Health School of Medicine, Farmington, CT
Correspondence: Zachary Stinson, MD, 13535 Nemours Parkway, Orlando, FL 32827. E-mail: [email protected]
Received: September 12, 2022; Accepted: October 2, 2022; Published: November 1, 2022
Volume 4, Number 4, November 2022
Abstract:
Pediatric orthopaedic surgical procedures require a thoughtful multimodal approach to perioperative pain management that maximizes both patient comfort and safety. Local infiltrative anesthesia (LIA) provides limited anatomic coverage over a relatively short length of time. In comparison, regional anesthesia can provide increased anatomic coverage over a longer duration by applying anesthetic medication directly around a targeted peripheral nerve or nerves. Peripheral nerve blocks (PNB) are commonly used in pediatric and adolescent sports medicine procedures, especially for anterior cruciate ligament reconstructions (ACLR). Yet there remains a wide variation in how PNBs are performed with limited evidence in support of their overall benefits in children relative to potential complications, such as prolonged sensory or motor nerve paralysis. Even less is known about their use and indications for a variety of non-sports pediatric orthopaedic procedures. This article provides a review on the uses and indications of both local and regional anesthesia in pediatric orthopaedics with a discussion on the available evidence in the literature on safety and efficacy.
Key Concepts
- The use of PNBs in the pediatric population has been increasing, especially for ACLR procedures.
- PNBs can result in improved immediate postoperative analgesia, but they have not been shown to significantly decrease overall postoperative opioid consumption.
- PNBs have a higher risk profile than LIA, and the incidence and long-term significance of these risks has not been sufficiently studied.
- The use of ultrasonography improves the safety and effectiveness of PNBs.
Introduction
The use of multimodal pain management in pediatric orthopaedic surgery requires a thorough understanding of the types of local and regional anesthetic options available and their application for various procedures. It is also important to recognize their limitations, especially in the pediatric population. The purpose of this article is to provide an overview of how peripheral nerve blocks are performed in addition to the various types of blocks and their respective indications (Table 1). It also reviews outcomes reported in the literature specific to the pediatric population. This content can potentially serve as a practical guide for optimizing the perioperative anesthesia plan for different pediatric orthopaedic procedures. While indications and techniques for regional anesthesia have evolved, knowledge surrounding the safety and efficacy lags behind. Sports procedures, especially anterior cruciate ligament reconstructions (ACLR), have received the most attention on the use of PNB in pediatric patients. DelPizzo found a trend towards an increase in the use of regional anesthesia (RA) in patients under the age of 21 in a retrospective review from 2005 to 2015, but the overall use of RA was below 25%.1 Factors associated with increased use included ACLR, increasing age, and sports injury. Hispanic ethnicity and Medicaid insurance were associated with decreased use of RA. Despite the increasing use of RA in ACLR, there is no consensus on the optimal type of PNB or if they provide substantial benefit over the potential risks. Other procedures in pediatric orthopaedics, such as congenital hand procedures or hip deformity procedures, have received considerably less attention with regard to the use of PNB. While small series have shown some promising outcomes on the use of PNB for these and other pediatric orthopaedic procedures, further investigation is warranted.
Table 1. Common Peripheral Nerve Blocks
| Common Peripheral Nerve Blocks | ||||
|---|---|---|---|---|
| Type | Nerve Distribution | Anatomic Location | Example Surgeries | |
| Upper Extremity | Interscalene | C3-C7 | Proximal Humerus | Shoulder Arthroscopy |
| Supraclavicular | C5-T1 | Clavicle Fracture ORIF | ||
| Axillary | Musculocutaneous, Median, Ulnar, Radial | Elbow, Forearm and Hand | Elbow Arthroscopy, Syndactyly Repair | |
| Lower Extremity | Femoral | Femoral Nerve | Anterior Knee and Lower Leg | Knee Arthroscopy/ACL/MPFL |
| Adductor Canal | Saphenous | Medial Knee and Leg | Knee Arthroscopy/ACL/MPFL | |
| Fascia Iliaca | Femoral Nerve and Lateral Femoral Cutaneous Nerve | Lateral Thigh and Hip | Hip Arthroscopy | |
| Sciatic | Sciatic Nerve | Broad Lower Extremity | Knee Arthroscopy/ACL/MPFL | |
| Popliteal | Tibial and Peroneal Nerves | Anterior, Lateral, Posterior Foot and Ankle | Ankle Arthroscopy, Ankle ORIF*, Syndactyly Repair | |
General Considerations
Location of Performing a PNB
In preparation for a surgical procedure, peripheral nerve blocks can be placed in the preoperative holding area or in the operating room. It is important to consider the potential benefits for the hospital system workflow when determining the location for performing blocks. Placement of the PNB in the preoperative area is particularly effective in the shared anesthesia care team model. For the first case of the day, and subsequently between cases, a PNB can be done by a dedicated regional anesthesia team in a specific PNB procedure room or in a preoperative bay, while a resident physician or mid-level anesthetist is available to prepare the operating room or take care of a prior patient. Additionally, earlier block placement allows time for the local anesthetic, especially more dilute solutions, to take effect. By the time that the operating room is ready for the patient and the patient has been transported to the operating room, the block is likely to have reached optimal analgesic effect. Moreover, the preoperative PNB can streamline subsequent workflow within the operating room and ultimately reduce time from patient entry into the operating room to surgical incision. Williams retrospectively studied the associations between anesthesia techniques and anesthesia-controlled time for a single knee procedure over a 3-year period.2 Consecutive patients receiving preoperative RA were associated with the lowest anesthesia control time, the lowest total turnover time, and the lowest incidence of unplanned hospital admission. On the other hand, there can be factors that complicate preoperative blocks. Some facilities may require that the surgical team re-evaluate the patient and complete certain tasks in the operating room which may require full function of the patient prior to PNB placement. Furthermore, most pediatric patients including adolescents need sedative medications to be administered in advance of the PNB. Such sedation requires a nurse to administer the medications, and intra- and post-procedure monitoring by nursing staff until departure to the operating room. This is an additional burden to nursing staff.
In the solo anesthesiologist model, a preoperative block could still be utilized but the advantages of performing the block in the operating room predominate. The redundancies required by preoperative blocks such as preoperative sedation, monitoring, and even microtasks such as disconnecting and reconnecting monitors when moving the patient from the preoperative area to the operating room are obviated. However, there remain spatial costs (e.g., ultrasound footprint), process costs (adjusting patient position to facilitate block placement), and time costs (performing a fractionated injection to reduce the risk of local anesthetic systemic toxicity). Additional equipment and space for performing the PNB remain within the operating room, which is already outfitted for procedures, eliminating the need for a designated room, space, or extra equipment. More importantly, if a complication or adverse event were to occur during or following the procedure, the operating room is a more optimal area to handle any emergency quickly, with more space and access to equipment that may not be as quickly or reliably available in a preoperative space.
Awake vs. Asleep
Due to developmental differences from adults, many children and adolescents may be less likely to tolerate regional anesthetic techniques without anxiolytics. Anesthesiologists can offer a range of sedation from mild sedation to general anesthesia. Historically, some blocks, particularly neuraxial techniques, were deemed safer with real-time feedback from the patient requiring little to no sedation for optimal patient engagement. However, studies show that cases of significant prolonged or permanent neurologic deficits after performing neuraxial procedures under general anesthesia are exceedingly rare,3 though this association should not be mistaken for causation.4 Patient engagement in peripheral nerve block procedures was a common technique perceived as an additional safety check. Anesthesiologists would confirm appropriate needle placement in PNBs with sensory feedback from the patient along with observed lack of sensory or motor changes following the procedure. Whether this approach truly improved the safety profile of PNBs has historically been debated. Much of this debate has been put to rest with the Pediatric Regional Anesthesia Network database. With over 100,000 regional anesthetics applied under general anesthesia, there were no reports of permanent neurologic damage, and the rate of transient neurologic deficit lasting less than 3 months was 2.4 per 10,000.5 In addition to the lack of evidence of increased risk of PNBs under general anesthesia, technological advancements and equipment sophistication have buttressed the safety profile of these procedures. Furthermore, the modern regional team can accomplish induction of general anesthesia and placement of a regional block expeditiously without interrupting flow of the operating room. This may also have additional institutional benefits in the realm of patient experience and patient satisfaction.
Nerve Stimulator vs. Ultrasound Technique
Purely anatomic landmark-based placement of nerve blocks has largely fallen out of favor in most settings. Technological advances improved appropriate needle placement, starting with nerve stimulation gaining significant clinical ground in the 1980s and 1990s. Ultrasound guidance for needle placement of peripheral nerve blocks started in the 1990s (Figure 1), but grew tremendously in the late 2000s and over the last decade has become commonplace. Ultrasound machines and probes have evolved to feature smaller footprints, battery power, more mobile platforms, and improved visualization through higher quality screens. Over the last several years, handheld ultrasound probes connecting to smartphones and tablets (Figure 2) have sprouted into the market at competitive prices which has lowered the barrier to entry for many settings and institutions and further led to the democratization and proliferation of point-of-care ultrasound use. The early literature comparing ultrasound and nerve stimulation showed superiority6 with ultrasound being associated with less procedural discomfort, less time to perform, and higher success rate.7 A meta-analysis of 15 randomized controlled trials (RCTs) of ultrasound vs. NS for nerve catheter placement showed higher success rates with ultrasound and less risk of vascular puncture.8 Ultrasonography has significantly decreased the risk of pneumothorax with supraclavicular block. Local anesthetic systemic toxicity (LAST) risk was also higher with nerve stimulation.9 Since it is a sensory nerve, the saphenous nerve block and adductor canal block are not typically done with nerve stimulation and rely on ultrasound guidance. While many practices are phasing out nerve stimulation in favor of ultrasound guidance, some still advocate a hybrid approach with ultrasound, because of its specificity in ruling out intraneural needle placement with low current stimulation <0.2 mA.
Figure 1. Adductor canal block using an in-plane approach performed in the operating room with the patient under general anesthesia with ultrasound guidance.

Figure 2. Butterfly iQ+ ultrasound probe connected to a smart device.

PNB Medications and Dosing
The most commonly used local anesthetics in the PNBs described in this article are the long-acting agents bupivacaine and ropivacaine. These medications belong to the amino amide group of local anesthetics which act via sodium channel blockade, blocking nerve function which includes sensations of pain, temperature, proprioception, touch, and skeletal muscle tone for approximately 10–12 hours which is optimal for perioperative and postoperative pain management.10
Bupivacaine was created in 1957 and has earned an enduring presence through the decades since. It was plagued by the specter of its CNS-toxic and cardiotoxic effects and many efforts were made to minimize its potential harm. Owing to this, multiple strategies were developed to mitigate its risk, from intermittent aspiration of syringe and monitoring of EKG during injection, addition of epinephrine or other supplements as a marker of intravascular administration, and slow fractionated injection. Levobupivacaine has also been used since it may have a more favorable therapeutic index than the racemic mixture. In the 1990s, the industry was revolutionized by the work of Guy Weinberg who developed lipid rescue, in which intralipid acts as a sink for intravascular bupivacaine and can allow for return of spontaneous circulation and improved outcomes. Since bupivacaine is generic and listed as an essential medicine by the World Health Organization, it is easily accessible across the globe.
Ropivacaine was developed and brought to market in the 1990s as an alternative to bupivacaine with less cardiotoxicity, though expert opinion on this topic varies. Since the cardiotoxicity differs due to physicochemical and stereoselective properties, ropivacaine is supplied as an S-(-)-enantiomer. Per its product insert, the addition of epinephrine does not prolong the duration of action or limit systemic absorption of ropivacaine. As with bupivacaine, unintentional intravascular injection or high systemic absorption of ropivacaine can lead to seizures, global CNS depression, and cardiovascular collapse.11
One method currently utilized in the operative setting to extend the duration of action of bupivacaine and ropivacaine is with adjuvant medications such as dexmedetomidine, dexamethasone, opioids, and epinephrine given locally as a mixture. Dexamethasone has also been shown to extend duration of action via systemic administration. These medications extend the effective time for peripheral nerve blocks and can be considered when evaluating patients’ analgesic needs with a “multimodal perineural analgesia” approach, paying attention to best individual patient practices.12
With the same goal to increase the duration of peripheral nerve block analgesia, liposome-encapsulated bupivacaine (Exparel) was developed with the aim of prolonging the duration of action through slow controlled release. It is approved for use in adults via brachial plexus injection for shoulder arthroplasty and rotator cuff repair, in children age 6 and above for soft tissue infiltration and has been used for other PNBs. Sustained levels of plasma bupivacaine are detected 72 hours after infiltration and 120 hours after brachial plexus injection, but local clinical effect does not meet the same duration. The preponderance of data has not shown a significant benefit to this strategy compared with placebo or conventional bupivacaine.13,14 After 10 years of use, it has not yet supplanted conventional bupivacaine or ropivacaine in routine use, and its definitive role in perioperative medicine is not yet clear.
Interscalene Block
Interscalene nerve block targets the roots (C5-7) and trunks of the brachial plexus, providing anesthesia to the upper arms and shoulder (Figure 3). It has been particularly effective for arthroplasty and rotator cuff repair in adult patients and is commonly used for shoulder arthroscopy in adolescent patients undergoing labral repair. Common side effects include hemi-diaphragmatic paralysis, hoarseness, and Horner’s syndrome, and patients should be advised of these prior to the procedure. The incidence of diaphragmatic paralysis declines when using less local anesthetic volume which can be facilitated by ultrasound. Rare complications include epidural and intrathecal injection, vascular trauma, and pneumothorax (See Interscalene Block Video).
Figure 3. Interscalene Nerve Block Anatomical Distribution.

Supraclavicular Block
The supraclavicular block has experienced a marked resurgence in use during the era of ultrasound guidance: since the brachial plexus, subclavian artery, rib, and pleura can all be visualized, the risk of pneumothorax declines precipitously with the aid of this technology. It addresses the divisions of the brachial plexus (Figure 4). There is also a likelihood of diaphragmatic paralysis, though less compared to interscalene block. It can also be used for shoulder surgery by injecting local anesthetic near the upper and middle trunk, though it can also be used for distal arm and hand by injecting near the middle and lower trunk structures. This block has tremendous versatility.
Figure 4. Supraclavicular Nerve Block Anatomical Distribution.
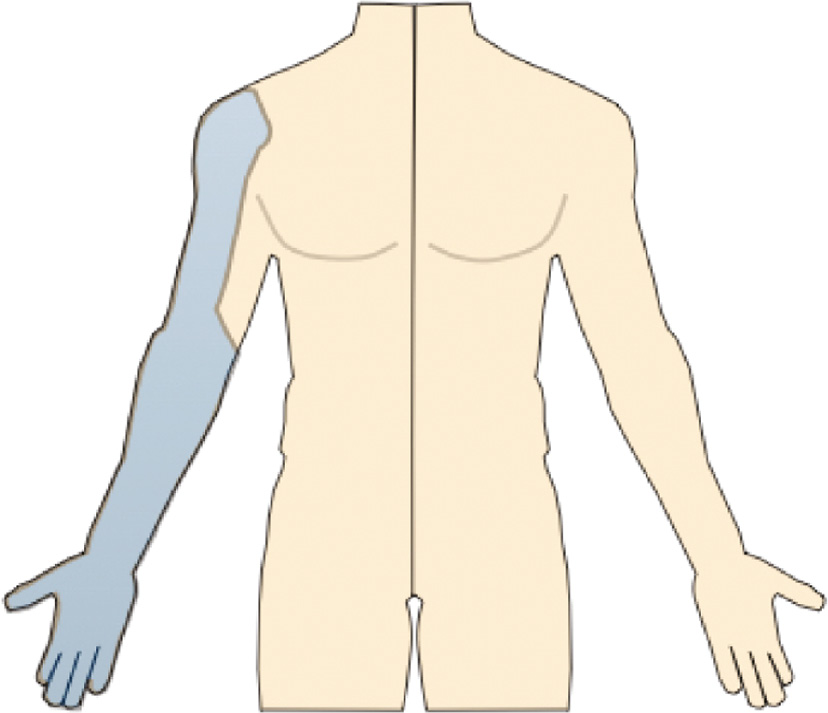
Infraclavicular Block
The infraclavicular block provides analgesia to the upper arm and elbow. It approaches the brachial plexus cords inferior and medial to the coracoid process (Figure 5). Further medial and deep run the axillary artery and vein. This block is especially useful for catheter placement in the posterior cord for major reconstructive procedures. Pneumothorax is also a risk with this procedure; thus, a lateral approach is typically utilized.
Figure 5. Infraclavicular Nerve Block Anatomical Distribution.

Axillary Nerve Block
In the adolescent and pediatric population, the axillary nerve block is popular for arm and hand procedures (Figure 6). Literature shows that with this approach however there is a 40-50% risk of missing the musculocutaneous nerve as it exits proximally from the axillary sheath. Thus, a separate block of the musculocutaneous nerve may also be done to ensure complete anterior forearm analgesia. For placement of this block in adults, the trans-arterial approach is commonly used; however, children have a greater incidence of vessel spasm and increased potential for ischemia with this approach compared to adults. To reduce the risk of these complications, this block is often performed with a single injection technique in children, which requires the use of nerve stimulation of the biceps for confirmation of proper needle placement.
Figure 6. Axillary Nerve Block Anatomical Distribution.

Sciatic Nerve Block
The sciatic nerve can be blocked in the subgluteal area for surgery involving the posterior thigh and knee or in the popliteal fossa (popliteal nerve block) for surgeries of the calf, ankle, and foot. Ultrasound can be used to visualize the sciatic nerve between the greater trochanter and the ischial tuberosity within the gluteus maximus muscle or in the popliteal fossa adjacent to the popliteal artery where it divides into the tibial and common fibular (peroneal) nerves (Figure 7). Complications include hematoma, risk of skin infection, and LAST (See Popliteal Sciatic Nerve Block Video).
Figure 7. Sciatic Nerve Block Anatomical Distribution.

Femoral Nerve Block
The femoral nerve block provides analgesia to the anterior thigh and knee. This block is useful for arthroscopic knee surgery. The femoral nerve is located within the inguinal crease lateral to the femoral artery (Figure 8). Due to the nerve’s proximity to major vasculature, vessel puncture is possible with risk of hematoma formation. Transient motor block of the quadriceps is expected, the long-term implications of which have been debated. In the short-term each patient needs to be on crutches or have a locked knee-immobilizer on until the block resolves.
Figure 8. Femoral Nerve Block Anatomical Distribution.

Fascia Iliaca Block
The fascia iliaca block is a plane block, aiming to block the femoral, obturator, and lateral femoral cutaneous nerves via cephalad spread of local anesthetic. It is intended as a simpler alternative to the lumbar plexus block. It can be used for hip arthroscopy and surgeries of the anterior thigh and knee (Figure 9). Use of dilute injectate may minimize motor block. It is performed in the supine position using ultrasound. The needle is introduced in-plane inferior to the inguinal ligament and guided beneath the fascia iliaca.
Figure 9. Fascia Iliaca Nerve Block Anatomical Distribution.

Adductor Canal
The adductor canal block (or saphenous nerve at the mid-thigh) has grown tremendously in use over the last decade, especially for ACLR, mainly due to the promise of reduced potential for motor block that plagues the femoral block. It requires ultrasound guidance and has been used for surgery of the knee primarily. The patient is positioned supine with the leg straight. The nerve is identified as it exits from the adductor canal adjacent to the femoral artery. As it is followed distally, it becomes more superficial, traveling with an arterial branch just deep to the sartorius muscle, and injection is done deep into the sartorius muscle at the lateral border of the artery (Figure 10).
Figure 10. Adductor Canal Nerve Block Anatomical Distribution.

Block Combinations
Combinations of lower extremity blocks are often utilized to maximize anatomic coverage. This includes a femoral block combined with a sciatic block, adductor canal block combined with a sciatic block, or an adductor or femoral block combined with an infiltration between the popliteal artery and the capsule of the knee (IPACK) block. Both the adductor canal block and IPACK block are favored due to their motor sparing approach.
Block Complications
The most common adverse events surrounding use of PNB include acute and prolonged motor weakness. Nerve injury following injection occurs with a reported incidence of 0.04%.15 As previously mentioned, postprocedural falls following femoral nerve block have prompted recommended use of a knee immobilizer postoperatively. The incidence of systemic toxicity secondary to intravascular injection is estimated at 0.03%. The use of ultrasound guidance has decreased the incidence of intravascular injection. A concern specific to interscalene and supraclavicular blocks is the potential for pneumothorax. As noted above, this risk has been shown to be significantly decreased by the use of ultrasound.
Local Infiltrative Anesthesia in Pediatric Orthopaedic Procedures
While the use of regional anesthesia has gained increased popularity for some pediatric orthopaedic procedures as described above, local infiltrative anesthesia (LIA) has remained a commonly used method of optimizing surgical site analgesia in pediatric orthopaedics. LIA has less potential for iatrogenic injury when compared to PNB, and various formulations of LIA are readily available and easy to administer with minimal set up and time required. Common anesthetics used include bupivacaine, ropivacaine, and lidocaine with dosing based on patient weight (Table 2). Bupivacaine is the most commonly utilized due to its longer half-life. These anesthetics can be used in conjunction with other medications including epinephrine, morphine, methylprednisolone, and/or clonidine creating an anesthetic cocktail (Table 3). Cocktails should be assessed for drug-drug interactions assessing how the half-lives, reactivity, and metabolism of individual components of the cocktail are affected when combined. LIA is contraindicated when patients are hypersensitive to local anesthetic drugs or components of the cocktail, have a history of coagulopathy, have cardiac impairment, have injection site infections, or are septic.
Table 2. Types of Local Anesthetic Medications and Dosing
| Generic | Brand Names | Maximal Dosing without Vasoconstrictors | Maximal Dosing with Vasoconstrictors | Onset Of Action (min) | Duration of Action | Toxicities /OD Complications |
|---|---|---|---|---|---|---|
| Bupivacaine | Exparel, Marcaine | 2.5 mg/kg per dose | 3 mg/kg per dose | 2-10 min | 120-175 min [w/epinephrine 180-480 min] |
Central nervous system dysfunction: tremors, conclusions, respiratory failure, and cardiovascular disturbances. |
| Lidocaine | Xylocaine | 4.5 mg/kg per dose | 7 mg/kg per dose | 1-3 min | 30-120 min [w/epinephrine 120-240 min] |
|
| Ropivacaine | Naropin | 2-3 mg/kg per dose | 3-4 mg/kg per dose | 3-15 min | 120-240 min [w/epinephrine 180-480 min] |
Table 3. LIA Cocktails Reported in the Literature
| Author | Procedure | LOE | Delivery Method | Cocktail | Location/Indication | Outcome |
|---|---|---|---|---|---|---|
| Lin et al. | Trigger Thumb Release | 1 | LIA | 2.5 ml of 0.25% Bupivacaine | Before surgical incision or immediately after skin closure | LIA before the incision is a low risk, easy method to reduce general anesthesia requirements and may decrease sevoflurane exposure more substantially in longer pediatric orthopaedic procedures |
| Tseng et al. | Displaced distal radius fracture | 2 | HB | Lidocaine of varying concentrations | N/A | HB treatment effect on pain severity was significantly better than PSA. No statistically significant reduction failure or adverse effects between HB and PSA groups. HB is a safe and effective alternative of anesthesia without inferior pain relief compared with PSA. |
| Astacio et al. | Supracondylar Humeral Fracture | 2 | HB | 0.25% Bupivacaine | Postoperative pain adjuvant | Similar morphine equivalent consumption and Face Pain Scale-Revised Survey results. Do not favor the use of HB to improve pain control and decreased need for opioid use on postoperative day 1. |
| Danielwecz et al. | Spinal Deformity Correction Procedure | 2 | LIA | 0.25% Bupivacaine | Postoperative pain control administered before wound closure | Overall reduction of opioid use on postoperative day 1. Decreased antiemetic requirement. Pain sensation moderately reduced up to 4 hours post-surgery in LIA patients. First morphine administration did not differ and increased postoperative wound bleeding was observed in LIA group. LIA with bupivacaine should not constitute a routine procedure. |
| Ross et al. | Posterior Spinal Fusion | 3 | CWI | 0.5% Bupivacaine (4 ml/h for 100 hours) | N/A | Overall reduction of opioid use of postoperative day 1. Decreased antiemetic requirement. |
| Reynolds et al. | Posterior Spinal Fusion | 3 | CWI | 0.25% Bupivacaine in sterile saline 4 ml/h for a total volume of 410 ml (102 hours) | Postoperative pain management | LIA decreased opioid consumption by 38% within 24 hours. No difference in incidence of adverse effects. |
| Wade et al. | Posterior Spinal Fusion | 3 | CWI | 0.25% bupivacaine at 4 ml/h for 72 hours | N/A | LIA reduced normal opioid requirements by 0.98 mg/kg and VA pain score by 0.67 points. |
| Bulut et al. | Extremity Surgery | 1 | WC | 0.2 mk/kg, 0.5% Bupivacaine | Prior to closure of fascial layer | LIA may provide long-term and efficient postoperative analgesia. |
| Herrera et al. | Isolated Femoral Fracture | 3 | HB | 0.5% Bupivacaine | N/A | HB increases time to first narcotic dose after surgery. HB is a simple, quick, and effective method of postoperative pain control after elastic femoral nailing in children. |
| Tran et al. | ACL Reconstruction | 1 | IA | (1 ml/kg 0.25%) Bupivacaine and Clonidine (1 μg/kg) and morphine (5 mg) | Prior to surgery in incision area | LIA patients required morphine PCA sooner and had increased nausea and vomiting incidence. After pediatric ACL reconstruction, FSNB with bupivacaine-clonidine provides better analgesia with fewer side effects than LIA with bupivacaine-clonidine-morphine. |
| Mitchell et al. | ACL Reconstruction | 3 | LIA | 5 mg of morphine and 10 mg of 0.2% ropivacaine diluted into 40 mL of lactated Ringer’s solution | Administered at the closure of all incisions. | No significant difference in MME consumption. LIA is a viable alternative for analgesia in pediatric patients undergoing BTB ACL reconstruction. |
| Schloss et al. | Arthroscopic Knee Surgery | 3 | IA | 0.25% bupivacaine (10 mL) | After arthroscopic knee surgery in pediatric patients, a FNB shortens hospital stay, reduces opioid requirements, and decreases postoperative pain scores. For anterior cruciate ligament repairs, FNB lowers postoperative admission rates. | |
| Novais et al. | Surgical Hip Displacement for FAI | 3 | LIA | 0.2% Ropivacaine 0.3 ml/kg (max 40 mg), morphine 0.1 mg/kg (max 5 mg), methylprednisolone 0.5 mg/kg (max 40 mg), and saline with epinephrine 1 mcg/ml, and infusion of ropivacaine 0.2% run at 6 ml/h until next morning postoperatively | Intraoperatively, after the hip capsule was closed and before the trochanteric osteotomy was fixed with screws | LIA was more effective at controlling pain 12 hours after surgery in comparison with PCA and had similar pain control to epidural. LIA was associated with significantly lower need for opioids and a shorter hospital stay compared with PCA and epidural protocols. LIA should be considered for pain management. |
| Pedersen et al. | Unilateral Hip Reconstruction (Cerebral Palsy) | 1 | LIA | Ropivacaine (2 mg/kg) and epinephrine (5 μg/mL) as infiltration and ropivacaine (0.5 mg/kg) as bolus. |
Intraoperative infiltration and boluses 8 hours and 20 hours postoperatively. | Mean level of pain 4 hours postoperatively was lower in the epidural group than in both the LIA group and the placebo group. Epidural analgesia is superior to LIA for early postoperative pain management in this patient population. |
| Schaver et al. | ACL Reconstruction | 3 | LIA | 300 mg of .5% ropivacaine, 30 mg of 30 mg/mL ketorolac, .6 mg of 1 mg/mL epinephrine, and 5 mg of .5 mg/mL morphine mixed in 100 mL .9% sodium chloride | Periarticular injection following closure of incisions with including both intraarticular injection and all sites of surgical exposure | Results of early pain scores, opioid consumption, and length of hospital stay were similar between the use of this LIA cocktail and ACB for ACLR. |
LIA is limited in its duration of analgesia as well as the anatomic location where analgesia is able to be achieved when compared to PNB. While PNB offers a theoretical advantage over LIA in regard to those limitations, there is not substantial clinical evidence that PNB provides significantly improved outcomes over LIA, especially in the pediatric population (Table 4). In addition, there are many clinical scenarios where PNB is contraindicated, such as in settings of trauma where there is the concern for the potential of compartment syndrome or the need of close neurologic monitoring. In contrast, LIA has very few contraindications and can provide a great adjunct to postoperative analgesia in a wide range of clinical settings.
Table 4. Studies Comparing LIA vs. PNB
| Study | Surgery | Anesthesia | LOE | N | Age | Hospital Stay | Postop Morphine | Units | VAS Score Postop |
|---|---|---|---|---|---|---|---|---|---|
| Tran et al. | ACL Recon | FSN-B | 1 | 16 | 15 (2) | 7 (13) | MME (mg) | 1.8 (3) | |
| Intraarticular Bupivacaine, Clonidine, Morphine | 18 | 15 (2) | 21 (21) | MME (mg) | 5.4 (3) | ||||
| P-Value | >0.05 | 0.03 | 0.0002 | ||||||
| Mitchell et al. | ACL Recon | FN-B | 3 | 58 | 16.4 (14.2-19.9) | 28.6 (15.2) | MME (mg) | 2.7 (1.6) | |
| Intraarticular Morphine and Ropivacaine Injection | 58 | 16.9 (14.1-20.2) | 24.4 (13.8) | MME (mg) | 3.0 (1.4) | ||||
| P-Value | 0.063 | 0.108 | 0.19 | ||||||
| Schloss et al. | Knee Surgery | FN-B | 3 | 131 | 14.8 (2.8) | 11.7 (8.1) | 80 (61%) | # Morphine Rescues | 4 (4) |
| Intraarticular Bupivacaine | 245 | 15 (2.8) | 15.8 (10) | 175 (71%) | # Morphine Rescues | 5.3 (3.1) | |||
| P-Value | 0.044 | 0.04 | 0.0004 | ||||||
| Novais et al. | Surgical Hip Dislocation for FAI | Epidural | 3 | 72 | 16.3 (2.5) | 72 (25-120) | |||
| LIA | 20 | 16.8 (2.1) | 38 (21-67) | ||||||
| IV PCA | 29 | 17.4 (2.3) | 61 (23-99) | ||||||
| P-Value | 0.1098 | ||||||||
| Pedersen et al. | Unilateral hip recon. | Epidural | 1 | 6 | 7.5 (5-11) | 0.63 (0.15) | Total Opioid Consumption (mg) | 1 (1) | |
| LIA | 5 | 8 (5-13) | 0.31 (0.17) | Total Opioid Consumption (mg) | 0.8 (1.8) | ||||
| Placebo | 6 | 6.5 (3-10) | 0.66 (0.42) | Total Opioid Consumption (mg) | 4 |
Reported Outcomes in Pediatric Orthopaedics
Elective Upper Extremity Procedures
Shoulder Arthroscopy
Edmonds et al. demonstrated effective use of LIA for shoulder arthroscopy in a study of 200 pediatric patients (mean age of 15.9 years). This study also compared the use indwelling glenohumeral catheter with local anesthesia injected peri-incisionally and intraarticularly.16 No chondrolysis was seen in the 15% of patients that had the intraarticular GH catheter, an important finding due to concerns brought forth by basic research suggesting deleterious effect on chondrocytes.17 While there are no studies specific to the pediatric and adolescent population on the use of PNB for shoulder arthroscopy, the use of interscalene blocks have been shown to provide improved pain management for shoulder arthroscopy in the adult population when compared to other pain relieving modalities. In the study by Edmonds et al., they reported that 51% of the patients had an interscalene block.16 They did report specifically on the pain management outcomes, but they did note that there were two patients with transient postoperative hand dysesthesias, two patients with postoperative headaches, and one patient with transient postoperative hypotension. It was not specified if those patients had received a PNB or LIA. In the systematic review by Warrender et al., complications associated with interscalene blocks included pneumothorax (6.1%), permanent nerve injury (<1%), and block failure (12.6 to 14%).18 Patients with interscalene blocks were also shown to have a significantly increased association with hemodynamic instability (P < .001), respiratory depression (P < .001), central nervous system impairment (P < .001), cardiac arrest (P < .001) seizures (P < .001) and death (P < .001). However, these findings were not the specific focus of the studies included in the systematic review and the small sample sizes limit the ability to draw definitive conclusions.
Hand Surgery
Thirty-five children undergoing radial club hand repair were studied by Ponde to evaluate the utility of an infraclavicular brachial plexus block. Intraoperatively, only one of 35 children showed reaction to pain, and in the postoperative period, 2/35 had high pain scores requiring tramadol. The authors concluded that this block provides satisfactory intra and postoperative analgesia.19
Lin et al. performed an RCT to determine if the timing of LIA (preincision vs. postrelease) of LIA affected postoperative pain or length of stay in pediatric unilateral trigger thumb release.20 The precision group comprised of six males and seven females with a mean age of 3.11±1.11 years, and the post incision group comprised of seven males and five females with a mean age of 3.19±0.95 years. They found that administering LIA pre-incision decreased cumulative sevoflurane dose, which can mitigate the risks of associated neurotoxicity, and there were no significant differences in postoperative pain scores, use of rescue pain medications, incidence of nausea, or time to discharge.
Elective Lower Extremity Procedures
Oberndorfer investigated the use of sciatic and femoral nerve blocks in 46 children undergoing lower extremity surgery. There were no block failures when ultrasound was used. Prolonged pain relief was achieved with smaller volumes of local anesthetic when compared to adults or blocks without the use of ultrasound.21
The following is a review of blocks specific for different regions.
Hip Reconstruction
Novais et al. assessed the efficacy of LIA vs. Epidural and Intravenous PCA after surgical hip dislocation (SHD) for the treatment of Femoroacetabular Impingement (FAI) in adolescents ranging from ages 12-21 (average age in PCA = 17.4+12.3; Epidural = 16.3±2.5; LIA = 16.8±2.1). The authors found that the LIA was more effective at controlling pain 12 hours postoperatively in comparison with PCA but had similar pain control to epidural. LIA was also found to be associated with significantly lower need for opioids and shorter length of hospital stay compared with the PCA and epidural protocols. The authors concluded that LIA should be considered for pain management after SHD for FAI treatment in adolescents.22
Pedersen et al. assessed the effect of epidural analgesia, LIA, and an approximated placebo on early postoperative pain in children with cerebral palsy who were undergoing unilateral hip reconstruction.23 The epidural group comprised of six patients with a mean age of 7.5 years (range: 5-11), the LIA group comprised of five patients with a mean age of 8 years (range: 5-13), and the placebo group comprised of 6 patients with a mean age of 6.5 years (range: 3-10). The authors found that epidural analgesia is superior to LIA for early postoperative pain management in this patient population. While the authors found that RA is superior, it is important to note the incredibly small sample size each group had in this study, and it would be important to look at this study in a large group.
Knee Arthroscopy and ACLR
Vorobeichik and colleagues conducted a three-part systematic review to determine the evidence surrounding use of PNB and LIA for patients undergoing ACLR. In part 1, the primary outcome was analgesic consumption. Eight RCTs met inclusion criteria with data from 716 patients included. The effect of adding a PNB varied between no improvement to modest benefits in opioid consumption. Adding a PNB to the use of LIA did not result in incremental analgesia. In part 2 of the series, the authors examined the evidence for using the adductor canal block (ACB) for analgesia after arthroscopic knee surgery, including ACLR. Postoperative rest pain severity score (VAS 0-10) at 6 hours after arthroscopic knee surgery was designated as the primary outcome. Data from 10 RCTs included 714 patients (358 intervention, 356 control). ACB marginally improved resting and dynamic pain up to 8 hours and reduced cumulative 24 hours analgesic consumption while preserving motor strength after simple knee arthroscopy. Administering ACB to patients undergoing arthroscopic ACLR did not improve pain control in the setting of multimodal analgesia.24–27 In part 3, the authors examined the evidence for using LIA after ACR. The primary outcome was cumulative analgesic consumption during the first 24 hours postoperatively. Eleven RCTs were included in the analysis. Use of LIA significantly reduced the cumulative 24-hour postoperative analgesic consumption when compared to controls. LIA also decreased the risk for opioid related side effects.
Maheshwer reported a meta-analysis of 27 level 1 RCTs comparing subjective pain scores, opioid consumption, and complications during the first 24 hours after primary ACLR in patients receiving spinal vs. regional blocks (FNB, ACB) vs. local injections.28 Pain control (VAS scores) after primary ACLR was significantly improved at 8 to 12 hours in patients receiving regional anesthesia as compared with spinal anesthesia. No significant difference in VAS was appreciated when spinal, regional, and local anesthesia groups were compared at 12-24 hours. Multiple additional studies had similar findings. In an investigation assessing pain control after ACLR with bone–patellar tendon–bone autograft using an FNB or local injection at wound closure, Mehdi reported no significant difference in VAS pain levels at 4 hours (P = .27) or 24 hours (P = .31) after surgery.29 Mayr found no significant difference in pain scores during the first 24 hours after surgery (157 patients) in a prospective randomized study comparing FNB and local intraarticular anesthesia administered preoperatively.30 Stebler compared the analgesic efficacy of periarticular LIA to regional ACB for isolated ACLR surgery and found similar early postoperative pain levels without impairing functional outcomes.31 Schaver conducted a retrospective review of prospectively collected data from an ACL registry to compare postoperative pain and recovery after anterior cruciate ligament reconstruction (ACLR) in patients who received an adductor canal block (ACB) or periarticular local infiltrative anesthesia (LIA).32 Postoperative pain levels and total opioid consumption and hospital recovery time were similar for LIA and ACB in the immediate postoperative period. The primary weakness among these studies is the lack of homogeneity among the anesthetic and analgesic protocols because each study introduces a unique multimodal pain regimen that consists of various anesthetic agents and adjunct local injections.
While the above studies do not provide strong evidence to support the routine use of PNB ACLR, there are several studies that demonstrate favorable outcomes of PNB in ACLR. In a 2005 RCT, Tran et al. assessed the analgesia and side effect profile of femoral-sciatic nerve block (FSNB) vs. intraarticular infiltration in 36 children (mean age 15±2) undergoing ACL reconstruction.33 The LIA group required more intraoperative fentanyl, morphine PCA sooner, and had increased side effect prevalence (vomiting). The authors concluded that FSNB provides better analgesia with fewer side effects than the LIA group. Schloss et al. retrospectively investigated the outcomes of pediatric patients receiving FNB in addition to general anesthesia for arthroscopic knee surgery compared with those receiving intraarticular bupivacaine (0.25%,10ml) and general anesthesia.34 The FNB group comprised of 131 patients with a mean age of 14.8 years (2.8), and the LIA group comprised of 245 patients with a mean age of 15 years (2.8). Those in the FNB group had shorter hospital stays, a reduction in opioid requirements (p = 0.04), and decreased postoperative pain scores. The authors also found that those undergoing ACLR with FNB had lower postoperative admission rates, which is similar to the findings in the study by Tran et al.
More recently, a study performed in 2021 by Mitchell et al. provides even further conflicting results. Femoral nerve blocks were compared against intraarticular injection with ropivicaine and morphine with a retrospective pediatric cohort undergoing ACLR.35 The FNB comprised of 27 males and 31 females with a mean age of 16.4 (range:14.2-19.9) and the LIA group comprised of 28 males and 30 females with a mean age of 16.9 (14.1-20.2). The authors could not identify a significant difference in morphine milligram equivalents (MME) consumed in the first 24 hours postoperatively and proposed that alternatively to Tran et al.’s finding that LIA is a viable alternative for analgesia in adolescent patients undergoing bone-patellar-tendon-bone graft ACLR. An additional prospective observation study on the use of light IV propofol anesthesia in conjunction with LIA in pediatric knee arthroscopy by Maldini et al. demonstrated the effective use of 15 mL of 2% lidocaine with epinephrine injected intraarticularly in 88 boys and 59 girls with a mean age 13.5±3.1 years.36 This combination for arthroscopic knee procedures provided effective sedation, good preservation of upper airway patency, rapid recovery, and pain relief without major side effects. The authors also noted that the majority of patients did not require postoperative analgesia. These contradicting findings highlight the need for more evidence on the role of PNB vs. LIA in pediatric and adolescent ACLR.
Foot and Ankle Surgery
In a 2001 survey to the members of the Pediatric Orthopaedic Society of North America regarding treatment approach to idiopathic clubfoot deformity, 39.4% of the tenotomy procedures via the Ponseti method were estimated to be performed under local anesthetic.37 Bor et al. noted at the end of Ponseti clubfoot percutaneous tenotomy, the surgical wound was infiltrated with 0.25% bupivacaine for postoperative pain relief in Israel, while in the U.S. acetaminophen rectal suppositories were administered.38 Due to other analgesic methods in this study and confounding variables, no conclusions could be drawn as to which postoperative analgesic methodology was more efficacious and further studies should be pursued.
Wejjakul et al.’s 2021 RCT compared the efficacy of a peripheral nerve block to LIA in pediatric foot and ankle tendon surgery.39 PNB and LIA provided comparable effective pain management in patients aged 1–6 years old, but interestingly, PNB showed significantly superior pain control in patients aged 7-15. These findings could indicate a difference in analgesia efficacy amongst different age cohorts even within the pediatric population, and further studies should investigate further age sub stratification in analgesic studies.
Pediatric Orthopaedic Trauma
Tseng et al. compared local hematoma blocks (HB) and procedure sedation and analgesia (PSA) for both adult and pediatric patients during reduction of displaced distal radial fractures to determine the level of pain relief, frequency of adverse effects, and reduction failure.40 They found that HB is a safe and effective alternative to PSA among pediatric patients (mean age of 10.3 years), and that there was no significant reduction failure between groups. They also noted HB treatment’s effect on pain severity as significantly better than PSA’s effect. Alternatively, in a prospective cohort study, Astacio et al. evaluated the efficacy of local 0.25% bupivacaine HB as postoperative pain control in pediatric patients (average age = 6 years, range 4-11) with supracondylar humeral fracture who underwent closed reduction pin fixation.41 This study’s results did not favor the use of HB to improve pain control or decreased opioid use on postoperative day 1. The HB group and control group showed similar morphine equivalent consumption and face pain scale-revised survey results. Of note, there were no HB associated complications reported, but the authors remained critical of HB use for pediatric supracondylar humeral fractures. The mixed results in the literature regarding the use of HB in upper extremity fractures indicates a need for more evidence to better understand their role in pediatric fracture management. The mean age difference in the studies described above may also have played a role in the differing findings, as some studies discussed later in this review have noted increased LIA efficacy as compared to regional anesthesia in children aged 1-6 years old when compared to children aged 7-15 years old.
Herrera et al. assessed the efficacy of hematoma blocks (HB ) in pediatric patients with isolated femoral shaft fractures treated with flexible elastic intramedullary nailing (IMN).42 The HB blocks group comprised of 12 males and one female with a mean age of 10.2 years while the control group comprised of 19 males and three females with a mean age of 9.7 years. The control group required more opioid administration within the first 2 hours postoperatively, and time to first rescue opioid dose postoperatively was over 5 hours earlier than the HB group. The authors concluded that HB is a safe and effective adjunct for pain control following elastic IMN for femoral shaft fractures.
In a systematic review of children with femoral shaft fractures, a fascia-iliaca compartment block was found to reduce the need for supplemental analgesia with few adverse effects. The authors concluded that use in the prehospital environment should be considered.43
Spine Surgery
Danielewicz et al.’s prospective cohort study assessed the effect of bupivacaine LIA administration prior to wound closure vs. no LIA in children and adolescents (8-17 years) subjected to spinal deformity correction procedure on postoperative opioid consumption, pain intensity, and bleeding in the early postoperative period.44 The study group was comprised of 13 females and four males with a mean age of 15.1 (range: 12.6-17.5), and the control group was comprised of 11 females and two males with a mean age of 14 (range: 8.5-17). Pain scores were slightly lower in the 0-4 hours period in LIA patients compared to the control group; however, after this time period no differences were observed. Increased bleeding was observed in the LIA group when compared to the control. This study concluded that despite the modest analgesic effects of LIA, due to the increased bleeding, LIA should not be part of pain control management in the pediatric population after spinal deformity correction.
Ross et al.’s 2011 retrospective analysis assessed whether an infusion of bupivacaine would reduce the need for IV opioids following posterior spine fusion.45 The study and control group’s mean ages were 14.1±1.8. The study group was comprised of 105 (81.4%) females and the control group was comprised of 91 (79.1%) females. Their data showed that continuous infusion of bupivacaine provided good analgesia with low pain scores and could have played a role in the significant reduction of the basal morphine use on postoperative day 1 by this group.45 Similarly, Reynolds et al.’s 2013 retrospective analysis of a patient with idiopathic scoliosis undergoing spinal fusion surgery found that the group undergoing continuous infusion of local anesthesia used less opioid analgesics during the first 24 hours after surgery.46 In Reynold et al.’s study, the study group comprised of 48 females and 24 males with a mean age of 13.9±2.2 years, and the control group comprised of 20 females and 5 males with a mean age of 13.2±2.2 years. Wade et al. corroborated these studies as well in their retrospective review of patient with adolescent idiopathic scoliosis undergoing posterior spinal fusion, finding that the use of a bupivicaine pump lowered pain scores, opioid requirements, and length of hospital stay.47 The study groups consisted of 108 females and 31 males with an average age of 14.4±1.7 years, and the control group consisted of 40 females and 17 males with an average age of 14.3±1.6 years.
Summary
With advancements in both technology and training associated with regional anesthesia combined with the emphasis on opioid-sparing multimodal perioperative analgesia, there has been an increase in the use of PNBs for pediatric orthopaedic procedures, especially in adolescent patients undergoing ACLR. While PNBs have shown improved immediate postoperative analgesia over LIA, there is no evidence of overall improvement in pain management and opioid consumption for the extended postoperative period. In addition, there has been limited reporting on the incidence of block-associated complications in the pediatric population. Further investigation is warranted on the use of PNBs in the pediatric population. Future studies should aim to standardize both the multimodal analgesic approach and the type of PNBs utilized among different pediatric orthopaedic procedures and include long-term prospective monitoring of both pain management and potential block-related complications. It is important to utilize a team-oriented approach to optimize the timing and location of PNBs, and ultrasound guidance should be utilized to maximize efficacy and safety.
Disclaimer
No funding was received for this study. The authors have no conflicts of interest to report.
References
- DelPizzo K, Fiasconaro M, Wilson LA, et al. The utilization of regional anesthesia among pediatric patients: a retrospective study. HSS J. 2020;16(Suppl 2):425–435. Epub 20201022.
- Williams BA, Kentor ML, Williams JP, et al. Process analysis in outpatient knee surgery: effects of regional and general anesthesia on anesthesia-controlled time. Anesthesiology. 2000;93(2):529-538.
- Allison CE, Aronson DC, Geukers VG, et al. Paraplegia after thoracotomy under combined general and epidural anesthesia in a child. Paediatr Anaesth. 2008;18(6):539-542.
- Raz A, Avramovich A, Saraf-Lavi E, et al. Spinal cord ischemia following thoracotomy without epidural anesthesia. Can J Anaesth. 2006;53(6):551-555.
- Walker BJ, Long JB, Sathyamoorthy M, et al. Complications in pediatric regional anesthesia: an analysis of more than 100,000 blocks from the pediatric regional anesthesia network. Anesthesiology. 2018;129(4):721-732.
- Danelli G, Fanelli A, Ghisi D, et al. Ultrasound vs nerve stimulation multiple injection technique for posterior popliteal sciatic nerve block. Anaesthesia. 2009;64(6):638-642.
- Bamgbade OA. Magnesium interscalene nerve block for the management of painful shoulder disorders. Saudi J Anaesth. 2018;12(2):343-345.
- Schnabel A, Meyer-Frießem CH, Zahn PK, et al. Ultrasound compared with nerve stimulation guidance for peripheral nerve catheter placement: a meta-analysis of randomized controlled trials. Br J Anaesth. 2013;111(4):564-572. Epub 20130605.
- Orebaugh SL, Kentor ML, Williams BA. Adverse outcomes associated with nerve stimulator-guided and ultrasound-guided peripheral nerve blocks by supervised trainees: update of a single-site database. Reg Anesth Pain Med. 2012;37(6):577-582.
- Shafiei FT, McAllister RK, Lopez J. Bupivacaine. StatPearls. Treasure Island (FL): StatPearls Publishing. Copyright © 2022, StatPearls Publishing LLC.; 2022.
- Graf BM. The cardiotoxicity of local anesthetics: the place of ropivacaine. Curr Top Med Chem. 2001;1(3):207-214.
- Kirksey MA, Haskins SC, Cheng J, et al. Local anesthetic peripheral nerve block adjuvants for prolongation of analgesia: a systematic qualitative review. PLoS One. 2015;10(9):e0137312. Epub 20150910.
- Ilfeld BM, Eisenach JC, Gabriel RA. Clinical effectiveness of liposomal bupivacaine administered by infiltration or peripheral nerve block to treat postoperative pain. Anesthesiology. 2021;134(2):283-344.
- Hussain N, Brull R, Sheehy B, et al. Perineural liposomal bupivacaine is not superior to nonliposomal bupivacaine for peripheral nerve block analgesia. Anesthesiology. 2021;134(2):147-164.
- Walker BJ, Long JB, De Oliveira GS, et al. Peripheral nerve catheters in children: an analysis of safety and practice patterns from the pediatric regional anesthesia network (PRAN). Br J Anaesth. 2015;115(3):457-462. Epub 20150722.
- Edmonds EW, Lewallen LW, Murphy M, et al. Peri-operative complications in pediatric and adolescent shoulder arthroscopy. J Child Orthop. 2014;8(4):341-344. Epub 20140601.
- Jansson H, Narvy SJ, Mehran N. Perioperative pain management strategies for anterior cruciate ligament reconstruction. JBJS Rev. 2018;6(3):e3.
- Warrender WJ, Syed UAM, Hammoud S, et al. Pain management after outpatient shoulder arthroscopy: a systematic review of randomized controlled trials. Am J Sports Med. 2017;45(7):1676-1686. Epub 20161013.
- Ponde V, Athani B, Thruppal S. Infraclavicular coracoid approach brachial plexus block for radial club hand repair. Paediatr Anaesth. 2007;17(9):863-866.
- Lin EE, Fazal FZ, Pearsall MF, et al. Local anesthetic injection before incision decreases general anesthesia requirements in pediatric trigger thumb release: a randomized controlled trial. J Pediatr Orthop. 2022;42(3):e285-e289.
- Oberndorfer U, Marhofer P, Bösenberg A, et al. Ultrasonographic guidance for sciatic and femoral nerve blocks in children†. Br J Anaesth. 2007;98(6):797-801.
- Novais EN, Kestel L, Carry PM, et al. Local infiltration analgesia compared with epidural and intravenous PCA after surgical hip dislocation for the treatment of femoroacetabular impingement in adolescents. J Pediatr Orthop. 2018;38(1):9-15.
- Kjeldgaard Pedersen L, Nikolajsen L, Rahbek O, et al. Epidural analgesia is superior to local infiltration analgesia in children with cerebral palsy undergoing unilateral hip reconstruction. Acta Orthop. 2016;87(2):176-182. Epub 20151106.
- Wei J, Yang HB, Qin JB, et al. Single-dose intra-articular bupivacaine after knee arthroscopic surgery: a meta-analysis of randomized placebo-controlled studies. Knee Surg Sports Traumatol Arthrosc. 2014;22(7):1517-1528. Epub 20130609.
- Bushnell BD, Sakryd G, Noonan TJ. Hamstring donor-site block: evaluation of pain control after anterior cruciate ligament reconstruction. Arthroscopy. 2010;26(7):894-900. Epub 20100513.
- Faunø P, Lund B, Christiansen SE, et al. Analgesic effect of hamstring block after anterior cruciate ligament reconstruction compared with placebo: a prospective randomized trial. Arthroscopy. 2015;31(1):63-68. Epub 20140917.
- Williams BA, Bottegal MT, Kentor ML, et al. Rebound pain scores as a function of femoral nerve block duration after anterior cruciate ligament reconstruction: retrospective analysis of a prospective, randomized clinical trial. Reg Anesth Pain Med. 2007;32(3):186-192.
- Maheshwer B, Polce EM, Paul K, et al. Regenerative potential of mesenchymal stem cells for the treatment of knee osteoarthritis and chondral defects: a systematic review and meta-analysis. Arthroscopy. 2021;37(1):362-378. Epub 2020/06/05.
- Mehdi SA, Dalton DJ, Sivarajan V, et al. BTB ACL reconstruction: femoral nerve block has no advantage over intraarticular local anaesthetic infiltration. Knee Surg Sports Traumatol Arthrosc. 2004;12(3):180-183. Epub 20040123.
- Mayr HO, Entholzner E, Hube R, et al. Pre- versus postoperative intraarticular application of local anesthetics and opioids versus femoral nerve block in anterior cruciate ligament repair. Arch Orthop Trauma Surg. 2007;127(4):241-244. Epub 20060523.
- Stebler K, Martin R, Kirkham KR, et al. Adductor canal block versus local infiltration analgesia for postoperative pain after anterior cruciate ligament reconstruction: a single centre randomised controlled triple-blinded trial. Br J Anaesth. 2019;123(2):e343-e349. Epub 20190524.
- Schaver AL, Glass NA, Duchman KR, et al. Periarticular local infiltrative anesthesia and regional adductor canal block provide equivalent pain relief after anterior cruciate ligament reconstruction. Arthroscopy. 2022;38(4):1217-1223. Epub 20211119.
- Tran KM, Ganley TJ, Wells L, et al. Intraarticular bupivacaine-clonidine-morphine versus femoral-sciatic nerve block in pediatric patients undergoing anterior cruciate ligament reconstruction. Anesth Analg. 2005;101(5):1304-1310.
- Schloss B, Bhalla T, Klingele K, et al. A retrospective review of femoral nerve block for postoperative analgesia after knee surgery in the pediatric population. J Pediatr Orthop. 2014;34(4):459-461.
- Mitchell BC, Siow MY, Pennock AT, et al. Intra-articular morphine and ropivacaine injection provides efficacious analgesia as compared with femoral nerve block in the first 24 hours after ACL reconstruction: results from a bone-patellar tendon-bone graft in an adolescent population. Orthop J Sports Med. 2021;9(3):2325967120985902. Epub 20210305.
- Maldini B, Miskulin M. Outpatient arthroscopic knee surgery under combined local and intravenous propofol anesthesia in children and adolescents. Paediatr Anaesth. 2006;16(11):1125-1132.
- Zionts LE, Sangiorgio SN, Ebramzadeh E, et al. The current management of idiopathic clubfoot revisited: results of a survey of the POSNA membership. J Pediatr Orthop. 2012;32(5):515-520.
- Bor N, Katz Y, Vofsi O, et al. Sedation protocols for Ponseti clubfoot Achilles tenotomy. J Child Orthop. 2007;1(6):333-335. Epub 20071103.
- Wejjakul W, Tangwiwat S, Pangthipampai P, et al. Does ultrasound-guided popliteal-sciatic nerve block have superior pain control in pediatric foot and ankle surgery? A randomized control trial. J Orthop Sci. 2022;27(4):844-849. Epub 20210526.
- Tseng PT, Leu TH, Chen YW, et al. Hematoma block or procedural sedation and analgesia, which is the most effective method of anesthesia in reduction of displaced distal radius fracture? J Orthop Surg Res. 2018;13(1):62. Epub 20180327.
- Astacio E, Echegaray G, Rivera L, et al. Local hematoma block as postoperative analgesia in pediatric supracondylar humerus fractures. J Hand Surg Glob Online. 2020;2(3):155-158. Epub 20200321.
- Herrera JA, Wall EJ, Foad SL. Hematoma block reduces narcotic pain medication after femoral elastic nailing in children. J Pediatr Orthop. 2004;24(3):254-256.
- Hards M, Brewer A, Bessant G, et al. Efficacy of prehospital analgesia with Fascia Iliaca compartment block for femoral bone fractures: a systematic review. Prehosp Disaster Med. 2018;33(3):299-307.
- Danielewicz A, Fatyga M, Starobrat G, et al. Subcutaneous bupivacaine infiltration is not effective to support control of postoperative pain in paediatric patients undergoing spinal surgery. J Clin Med. 2021;10(11). Epub 20210529.
- Ross PA, Smith BM, Tolo VT, et al. Continuous infusion of bupivacaine reduces postoperative morphine use in adolescent idiopathic scoliosis after posterior spine fusion. Spine (Phila Pa 1976). 2011;36(18):1478-1483.
- Reynolds RA, Legakis JE, Tweedie J, et al. Postoperative pain management after spinal fusion surgery: an analysis of the efficacy of continuous infusion of local anesthetics. Global Spine J. 2013;3(1):7-14. Epub 20130302.
- Wade Shrader M, Nabar SJ, Jones JS, et al. Adjunctive pain control methods lower narcotic use and pain scores for patients with adolescent idiopathic scoliosis undergoing posterior spinal fusion. Spine Deform. 2015;3(1):82-87. Epub 20141218.
