Current Concept Review
Perioperative Blood Pressure Management for Patients Undergoing Spinal Fusion for Pediatric Spinal Deformity
1Children’s Healthcare of Atlanta/Emory University Department of Orthopaedics, Atlanta, GA; 2British Columbia Children’s Hospital, Vancouver, Canada; 3Boston Children’s Hospital, Boston, MA; 4Stanford University, Palo Alto, CA; 5Children’s Hospital of the King’s Daughter, Norfolk, VA; 6Rainbow Babies and Children’s Hospital/Case Western Reserve School of Medicine, Cleveland, OH
Correspondence: Nicholas D. Fletcher, MD, Children’s Hospital of Atlanta, CPG Orthopedics, 1400 Tullie Rd. NE, Atlanta, GA 30329. E-mail: [email protected]
Received: October 11, 2022; Accepted: November 29, 2022; Published: February 1, 2023
Volume 5, Number 1, February 2023
Abstract
Posterior spinal instrumentation and fusion has become the gold standard for the definitive management of children and adolescents with spinal deformity. Despite continued innovations designed to improve the safety profile of this complex surgical undertaking, spinal cord injury and resulting loss of neurologic function remain a rare but devastating risk. The increasing power of instrumentation combined with more aggressive correction strategies puts the spinal cord at particular risk due to traction. While the surgeon has the luxury of complex neuromonitoring techniques to alert the team in the presence of a neurologic change during surgery, maintenance of spinal cord perfusion throughout surgery and in the early postoperative period should be considered to avoid spinal cord ischemia as it accommodates to its new position after deformity correction. This manuscript represents recommendations of the POSNA Quality, Value, and Safety Spine Committee for optimization of blood pressure in the perioperative period.
Key Concepts
- Surgeons should take an active role in establishing blood pressure parameters in patients undergoing spinal surgery in order to optimize spinal cord perfusion during all phases of care.
- Spinal cord perfusion is critical during all portions of patient care and thus thoughtful blood pressure monitoring should occur postoperatively as well as intraoperatively.
- Even relatively brief periods of hypotension may result in significant spinal cord ischemia.
Introduction
Modern spinal deformity surgery, whether using growth-friendly instrumentation for early onset scoliosis or posterior spinal fusion (PSF) for adolescent idiopathic scoliosis (AIS), relies heavily on posterior-based implants that work through some level of spinal column lengthening. Surgeons continue to innovate on the classic techniques of posterior thoracic pedicle screw instrumentation using flexible drill bits, patient-specific drill guides, intraoperative navigation, and robotics to improve the accuracy of screw placement. These techniques, in addition to increasing familiarity with free-hand techniques by even young surgeons, have led to an ever-decreasing rate of spinal cord injury (SCI) related to screw misplacement. Skaggs et al. reviewed over 22,000 pedicle screws placed using a flexible drill bit and found a 0.03% rate of screw misplacement requiring revision.1 Baky compared 112 patients with AIS who underwent PSF using a fluoroscopically assisted approach to 107 patients managed with the assistance of a CT-based navigation system and found a 3.6% incidence of screw revision in the fluoroscopically assisted technique compared to 0% in the navigation-based group.2 Gonzalez et al. evaluated 40 patients undergoing robotically assisted pedicle screw placement and found a 98.7% accuracy rate with no neurological injuries.3 Despite these technical advances, the surgeon should always remember that this triumph of technology often results in stretching the spinal cord beyond its limits as implantation of promising new instrumentation can have devastating consequences. Hamilton et al. reviewed 108,419 reports from the Scoliosis Research Society’s (SRS) morbidity and mortality database and documented the incidence of neurologic deficits including those from the nerve root, cauda equina, and spinal cord levels. This study found that 10.6% of new deficits failed to show any recovery postoperatively.4
Hypotensive anesthesia was long espoused for minimization of intraoperative blood loss during spinal surgery. Adjuncts to limit blood loss such as antifibrinolytics (i.e., tranexaemic acid), bipolar sealer devices, ultrasonic bone scalpels, cell salvage, and power instrumentation have likely decreased the need for these potentially risky anesthetic practices.1,5–10 The authors believe that other than during exposure, hypotensive anesthesia should be avoided in modern spinal deformity surgery. Recent studies have shown dramatically lower blood loss during spinal surgery than even 15–20 years ago, likely a result of both better techniques and improved technologies.5,9 Clearly, optimizing safety during instrumentation is paramount to a neurologically safe surgery; however, the surgeon should remember that spinal cord perfusion is equally critical to a successful outcome when compared to the technical aspects of implant placement. Yang and colleagues reviewed patients treated with PSF for AIS. Of the 452 patients enrolled, there were 30 neuromonitoring alerts intraoperatively. Twenty percent of these resolved with an increase in mean arterial pressure (MAP), belying the importance of spinal cord perfusion during correction manuevers.11 Both Vitale et al.12 and Lenke et al.13 have published intraoperative checklists to guide surgeons in the face of neuromonitoring changes in AIS as well as more complex spinal deformity. One of the primary responses to these changes in both checklists is to increase MAP.
Perhaps even more disturbing than the intraoperative loss of neuromonitoring data related to spinal cord hypoperfusion are the variety of case reports and small case series describing delayed paralysis in patients with an otherwise successful spinal fusion. Indeed, Welling et al.,14 Kia et al.,15 Quinonez et al.,16 Lovi et al.,17 and Chang et al.18 all describe cases of otherwise routine surgery with delayed neurologic changes from 3 to 60 hours postoperatively. While some authors documented a period of transient hypotension, others demonstrated a significant anemia (i.e., hematocrit of 20%) which may exacerbate an otherwise borderline hypotensive situation.17 The majority of these published accounts fortunately resulted in complete neurologic recovery, the authors know of a number of unpublished cases where neurological losses were permanent. Alam et al.19 published an even more concerning series of eight patients with both AIS and neuromuscular spinal deformity with a delayed quadriplegia occurring postoperatively. All patients had a cervical spinal cord ischemic lesion despite more distally located instrumentation in the thoracic spine. While the authors posit some concerns about the risk of a larger curve, more aggressive correction maneuvers, and the presence of cervical kyphosis in these cases as causative factors, it should be remembered that relative spinal cord hypoperfusion is the common feature of any ischemic event. Optimizing blood pressure, both intraoperatively and postoperatively, may minimize this risk. This paper seeks to define recommendations for surgeons to minimize the risk of spinal cord ischemia. It should be noted that these are simply recommendations of the POSNA Spine QSVI Committee and that there is extremely limited research to substantiate these suggestions. Our hope is to generate an awareness of delayed neurologic changes after spinal fusion.
Preoperative
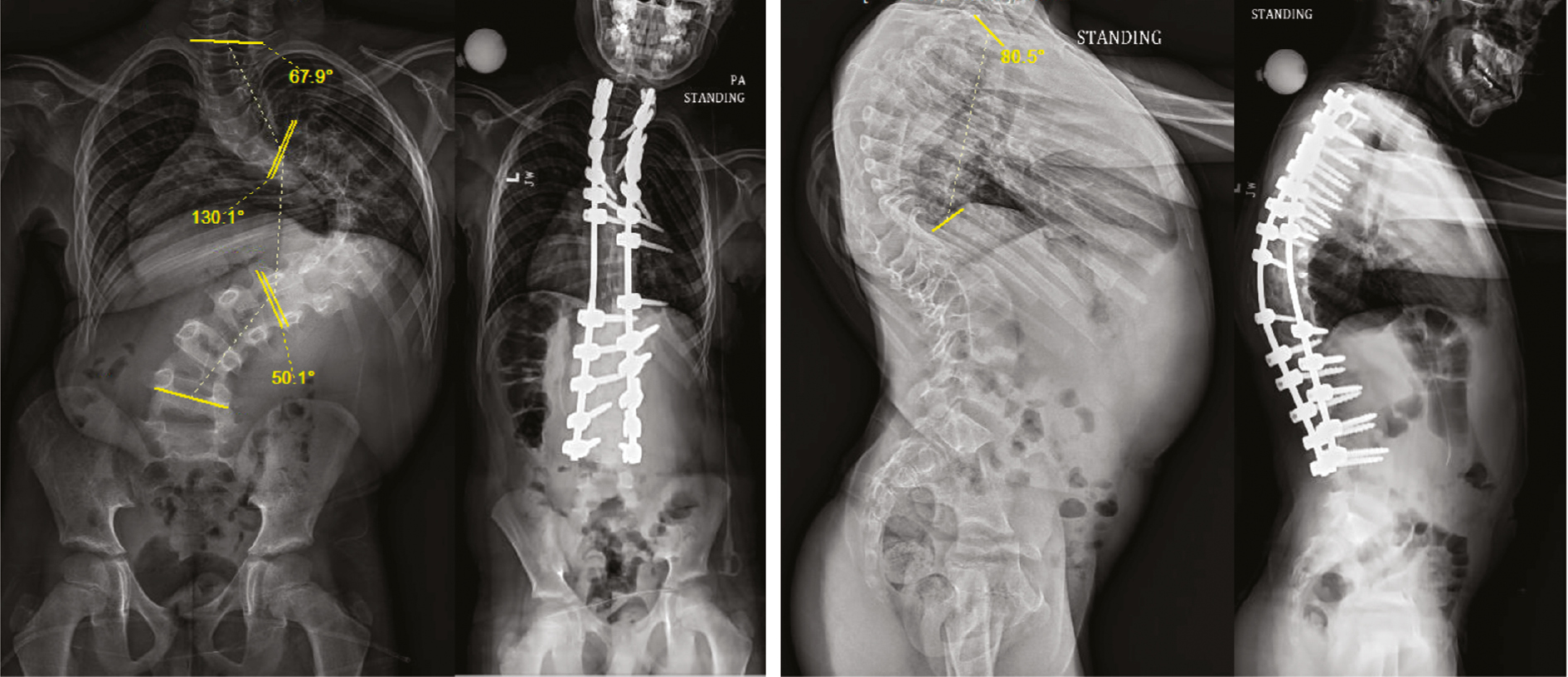
Outside of assessing for preexisting hypotension or anemia which may necessitate pharmacological support or earlier allogenic transfusion, the most important preoperative intervention may be in the planning for deformity correction. Prior planning will always prevent poor performance in the operating room and some forethought about surgical risk factors for spinal cord hypoperfusion can optimize outcomes. We recommend evaluating correction strategies, especially in larger and stiffer curves. Wang et al. have defined the deformity angular ratio (DAR), defined as the curve magnitude divided by the number of levels over which the curve is measured as a risk factor for IONM alerts. Patients with a total DAR (T-DAR), defined as the sagittal (S-DAR) plus the coronal (C-DAR) over 25 or a S-DAR over 15 were more likely to have loss of IONM data intraoperatively (Figure 1).20,21
Figure 1. An 11-year-old boy with severe juvenile idiopathic scoliosis. His Coronal-Deformity Angular Ratio (C-DAR) was 131°/7 levels = 18.7 and his sagittal-DAR (S-DAR) was 80.5°/10 levels = 8.1 giving him a T-DAR of 26.8. He had a temporary decrease in nMEP during correction which responded to removal of apical screws and recontouring the convex rod.
Sielatycki et al. classified the morphology of the spinal cord within the spinal canal based on the presence of cerebrospinal fluid (CSF) between the neural elements and the pedicle wall as well as any deformity imparted by this pedicle on the spinal cord. Type 3 spinal cords, defined as those with no CSF between the spinal cord and pedicle and deformity/flattening of the cord, were found to have a 28-fold higher rate of losing IONM data intraoperatively (Figure 2).22
Figure 2. Cross sectional T2 weighted MRI of the apical vertebrae from the child in Figure 1. The spinal cord is compressed against the pedicle wall with no intervening CSF. There is no significant flattening of the cord, consistent with a type 2 spinal cord.

Larger kyphosis also increases the possibility of neurological injury, especially in the setting of congenital curves or revision surgery.20,23–26 Larger curves may benefit from preoperative halo-gravity traction to allow the spinal column, including the spinal cord, to undergo slow and gently stretching while the patient is awake and can provide regular neurological examinations27 (Figure 3).
Figure 3. (A) A 12 year old male with Soto syndrome, a coronal curve of 113°, sagittal curve of 103° and a T-DAR of 24.2 (C-DAR = 11.3 S-DAR = 12.9). (B) He was treated with 6 weeks of halo gravity traction, taking his coronal curve down to 84° and his sagittal curve to 68°. (C) He was then treated with PSF without IONM changes.

A comparable strategy in more severe deformities is to place implants and apply temporary distraction with plans for staging the correction.28 While posterior column osteotomies (PCO) have increased in popularity,29 they have been associated with more IONM alerts, possibly due to the association with more aggressive corrections.30,31 Surgeons should anticipate IONM alerts during deformity correction in larger curves with multiple PCO’s and ensure that blood pressures are elevated to maximize perfusion of the spinal cord. The desire to maximize the power of modern instrumentation has often resulted in impressive radiographic outcomes following PSF however a neurologically devastated patient will never appreciate how straight their spine appears. Preoperatively defining the goals of surgery as achieving spinal balance, optimizing sagittal profile, and stopping curve progression can put the goal of surgery in perspective. Curve deformity correction is often the icing on the cake for these patients as limited data has correlated clinical improvements with minor differences in residual curve magnitude.32
Intraoperative
Modern spinal deformity correction mandates real time blood pressure monitoring using an arterial line. Non-invasive blood pressure monitoring, although a viable back-up in the setting of an unreliable arterial line, should not be used as the primary form of measuring mean arterial blood pressures. While not used on routine cases, surgeons may consider the utility of a central line in cases where monitoring cardiac function and output may be relevant such as patients with a cardiac history. As blood pressure management may require both volume and pharmacologic interventions, two large-bore (i.e., 18-gauge) intravenous lines should be established. Blood pressure can be augmented by vasopressors such as norepinephrine or dopamine. Norepinephrine has been shown to maintain mean arterial pressure with a lower intrathecal cerebrospinal fluid pressure and higher spinal cord perfusion pressure when compared to dopamine.32 The surgeon should also be aware of the presence of oversedation of the patient, often by a well-meaning but underexperienced anesthesiology resident, with high doses of propofol impacting initial IONM signals or undersedation with resulting hypertension upon incision which is quickly counteracted by a large bolus of opioids or anesthetic. An effective strategy is to carefully titrate each patient to a MAP goal upon initiation of surgery using an antihypertensive calcium channel blocker such as nicardipine for hypertension and vasoactive pressors for hypotension. A similar scenario can occur at the end of the surgery if anesthetics are stopped too early resulting in hypertension. The anesthesiologist should carefully balance increasing sedation in this scenario with the risk of creating hypotension and may consider the use of a bispectral index monitor to help measure the depth of sedation.33
Perhaps the most important time at which the surgeon can communicate the desired mean arterial pressure goals for the different portions of the surgery is during the surgical time out. While some feel that time outs should only incorporate the most critical aspects of the surgical case (i.e., allergies, site of surgery),34 the authors would contend that establishing blood pressure goals at the onset of surgery is as important as any of these. We typically state blood pressure goals for each component of the surgery (i.e., “MAPs 65-75 mmHg during exposure and 75-85 during instrumentation, PCO’s, and correction with maintenance at this level until the wound is closed”). Instituting these on every case and asking for confirmation creates an expectation that this is a critical component of surgery and establishes a pattern for the anesthesia team. After this has occurred regularly, it is typical that the anesthesiologist will preemptively ask for MAP parameters before surgery is started. The surgeon should also ensure that these constraints are conveyed to other members of the anesthetic team providing breaks to the primary team. In addition to establishing blood pressure goals, our typical practice is to also discuss the use of antifibrinolytics, the type of anesthesia (i.e., total intravenous anesthesia or TIVA), and the anticipated need for blood products (i.e., packed red blood cells, platelets, fresh frozen plasma, etc.).
Ideal blood pressure goals are surgeon and case specific; however, we will define our preferences here, realizing that there is no published data to support or refute their utility. They are provided as a guide and the authors would encourage surgeons to initiate a discussion with the anesthesia team to establish parameters at their own center. For typical adolescents with AIS, our goals as stated above are for MAP to fall around 65 mmHg during exposure and 75-85 mmHg during instrumentation and correction. These numbers increase for more complex cases (i.e., vertebral column resection) when we aim for a MAP between 75-85 mmHg during exposure but keep it above 85 mmHg during instrumentation and correction. These numbers may be even higher in the presence of myelopathy. While blood loss may be greater at higher blood pressures, even in the presence of antifibrinolytics,35 the use of cell salvage and multiple newer surgical instruments (i.e., bone scalpels) may act to counter massive hemorrhage.5,7–10 A unique difficulty that has not been previously discussed in the literature is the challenge of establishing levels of relative hypertension for cord perfusion in younger and smaller children. In consultation with hospital intensivists, the recommendations in Table 1 may help guide surgeons caring for younger children in the setting of high-risk deformity correction or true IONM changes. Assuming that blood pressure goals for a 15-year-old female weighing 75 kg undergoing PSF for AIS should be the same as for a 5-year-old female weighing 20 kg who is undergoing a hemivertebrectomy is likely incorrect. Adoption of a MAP of 85 mmHg in the setting of a IONM change likely puts the child at risk for intracranial hemorrhage. We therefore target a range of 20-25% above the child’s preoperative value during instrumentation and correction in younger children.
Table 1. Mean Arterial Pressure Goals for Children with Intraoperative Neuromonitoring Changes, either Temporary or Permanent, Based on Age
| Age | Mean Arterial Pressure Goal |
|---|---|
| 1–4 years | 70-80 mmHg |
| 5-9 years | 75-85 mmHg |
| 10-14 years | 80-90 mmHg |
| 15 years and older | 85-95 mmHg |
Postoperative
Surgeons should remember that the postoperative period for children undergoing spinal deformity surgery really commences as soon as the surgical incision is closed, as this typically represents the period when IONM no longer exists and neurologic assessment is purely clinical. Reacting to a neurologic change at any point thereafter often results in either emergent imaging or return to the operating room. As such, maintenance of appropriate blood pressures should continue while the patient wakes enough to provide an adequate examination.36 One of the biggest challenges for the surgeon at the conclusion of the surgery is the loss of relative control over patient blood pressure. During surgery, blood pressure management is a coordinated dance between the patient, the surgeon and the anesthesiologist. Once the child leaves the operating room, a variety of less informed and perhaps inexperienced team members may be monitoring the blood pressure and fail to appreciate the importance of spinal cord perfusion. The surgeon should strive to touch base with every team member, from the postoperative anesthesia nurse to the floor nurse, in any child at risk for spinal cord hypoperfusion.
While some centers still require all spinal deformity patients to be transferred to an intensive care unit setting,37 there is evidence that this is not necessary if appropriate staffing exists.38–41 Most centers currently send very few patients with AIS to the PICU and some centers have moved towards only requiring intensive care for select patients with neuromuscular scoliosis.42–44 It is our belief that any patient with a neuromonitoring change intraoperatively, even those that are short and transient, be admitted to the PICU for a minimum of 24 hours of observation. A change in IONM data suggests that the spinal cord is sensitive to the correction just imparted on it and that hypotension may push it over the edge to a point of inadequate perfusion. Patients with true neurologic deficits are obviously admitted to the PICU for monitoring and blood pressure management as well.
Patients with a temporary neurological change (i.e., decrease in motor evoked potentials during rod insertion which resolves with under correction of the rod and elevated MAPs) are admitted to the PICU. Our MAP goals are as above in Table 1 and surgeons should establish that these will be maintained for a minimum of 24 hours pending a stable neurological exam. One of our centers has further developed a specific note in our medical record that documents the intraoperative neuromonitoring change event and establishes our blood pressure goals in the medical record to minimize any ambiguity about the plan (Figure 4).
Figure 4. Example of standardized note in patients sustaining an intraoperative neuromonitoring change.
The surgeon should meet with the nurses and intensivist in a face-to-face manner to discuss MAP goals. While well meaning, it is likely that the intensive care team may not understand the importance of maintaining elevated MAP in an at-risk spinal cord. Temporary hypotension, perhaps seemingly harmless to the intensivist or bedside nurse, has been shown to provide an adequate window for spinal cord hypotension. Quinonez et al. reported a case of a child with a 125-degree scoliosis who was placed in the PICU with strict MAP parameters.16 A subsequent ascending paraplegia resulted in a rapid return to the OR for implant removal. A careful chart review revealed a short (~2 hour) period of relative hypotension which was felt to have caused this neurologic change. Hwang et al., using a rat model, found that 9 minutes of spinal cord ischemia resulted in mild, reversible spinal cord changes while 10 minutes of ischemia was the upper limit for moderate, reversible changes. All animals with 11 minutes or longer of spinal cord ischemia sustained a permanent, complete paraplegia.45 While no published human clinical data exists to add further support a specific time to permanent paraplegia, our anecdotal experience is that even a short period of hypotension can result in devastating spinal cord injury. The differences between this protocol and that used in the setting of a true neurologic change is simply the duration. The authors, while acknowledging the lack of supporting data, prefer to keep MAP goals in place in these patients for a minimum of 48-72 hours depending on the situation and may extend this in certain situations when postoperative examination may be less reliable (i.e., developmental delay, intubated patient, or a variable neurological exam). Central venous access is typically needed in these situations due to concerns for prolonged vasopressor use through a peripheral IV.
Perhaps one of the greatest challenges with postoperative blood pressure management is that most patients will spend some time on a general surgical floor with less frequent neurological checks and no ability to provide pharmacologic blood pressure support. It is our experience that floor nurses who may not be as well versed in the importance of blood pressure management in the postoperative setting may allow a MAP in the low 50s or even 40s to persist as long as the patient appears “well.” In an attempt to minimize ignoring hypotension in well appearing patients, one of us worked with our informatics team to create an adjunct to our standard postoperative electronic order set for otherwise uneventful spinal deformity surgeries. This order requires that nurses contact the house staff on call for any MAP under a certain threshold, typically 65 mmHg, with the expectation that a fluid challenge can be ordered. If a bolus is given without any response, then the patient’s hemoglobin and hematocrit are checked with a transfusion given if below a certain threshold based on the attending surgeon’s preference. If the hypotension persists, then the patient could be considered for transfer to the PICU for closer monitoring and vasopressive support. Three years after implementation of this protocol, the author is unaware of any patient who did not respond to volume resuscitation and required transfer to the PICU.
Conclusions
Blood pressure management in patients undergoing correction for spinal deformity should not be reserved for the anesthesia team and intensivists. Monitoring spinal cord perfusion in the setting of deformity correction falls on the entire team and the surgeon should play an integral role in assuring this. Having a preoperative plan to optimize blood pressure management, an intraoperative approach to maintenance of cord perfusion during deformity correction, and a postoperative strategy to minimize the risk of hypotension can potentially save a spinal cord at risk.
*POSNA QSVI Spine Committee: James T. Bennett, MD; Laurel C. Blakemore, MD; Nicholas D. Fletcher, MD; Lorena Floccari, MD; Ravi Ghag, MD, FRCSC; Michael P. Glotzbecker, MD; Daniel J. Hedequist, MD; Meghan N. Imrie, MD; Megan Johnson, MD; Selena Poon, MD; Peter F. Sturm, MD
Disclaimer
No funding was received. The authors report no conflicts of interest related to the manuscript.
References
- Skaggs DL, Compton E, Vitale MG, et al. Power versus manual pedicle tract preparation: a multi-center study of early adopters. Spine Deform. 2021;9(5):1395-1402.
- Baky FJ, Milbrandt T, Echternacht S, et al. Intraoperative computed tomography-guided navigation for pediatric spine patients reduced return to operating room for screw malposition compared with freehand/fluoroscopic techniques. Spine Deform. 2019;7(4):577–581.
- Macke JJ, Woo R, Varich L. Accuracy of robot-assisted pedicle screw placement for adolescent idiopathic scoliosis in the pediatric population. J Robot Surg. 2016;10(2):145-150.
- Hamilton DK, Smith JS, Sansur CA, et al. Rates of new neurological deficit associated with spine surgery based on 108,419 procedures: a report of the scoliosis research society morbidity and mortality committee. Spine (Phila Pa 1976). 2011;36(15):1218-1228.
- Bartley CE, Bastrom TP, Newton PO. Blood loss reduction during surgical correction of adolescent idiopathic scoliosis utilizing an ultrasonic bone scalpel. Spine Deform. 2014;2(4):285-290.
- Garg S, Thomas J, Darland H, et al. Ultrasonic bone scalpel (USBS) does not reduce blood loss during posterior spinal fusion (PSF) in patients with adolescent idiopathic scoliosis (AIS): randomized clinical trial. Spine (Phila Pa 1976). 2021;46(13):845-851.
- Gordon ZL, Son-Hing JP, Poe-Kochert C, et al. Bipolar sealer device reduces blood loss and transfusion requirements in posterior spinal fusion for adolescent idiopathic scoliosis. J Pediatr Orthop. 2013;33(7):700-706.
- Hardesty CK, Gordon ZL, Poe-Kochert C, et al. Bipolar sealer devices used in posterior spinal fusion for neuromuscular scoliosis reduce blood loss and transfusion requirements. J Pediatr Orthop. 2018;38(2):e78-e82.
- Mankin KP, Moore CA, Miller LE, et al. Hemostasis with a bipolar sealer during surgical correction of adolescent idiopathic scoliosis. J Spinal Disord Tech. 2012;25(5):259-263.
- Wang X, Sun G, Sun R, et al. Bipolar sealer device reduces blood loss and transfusion requirements in posterior spinal fusion for degenerative lumbar scoliosis: a randomized control trial. Clin Spine Surg. 2016;29(2):E107-E111.
- Yang J, Skaggs DL, Chan P, et al. Raising mean arterial pressure alone restores 20% of intraoperative neuromonitoring losses. Spine (Phila Pa 1976). 2018;43(13):890-894.
- Vitale MG, Skaggs DL, Pace GI, et al. Best practices in intraoperative neuromonitoring in spine deformity surgery: development of an intraoperative checklist to optimize response. Spine Deform. 2014;2(5):333-339.
- Lenke LG, Fano AN, Iyer RR, et al. Development of consensus-based best practice guidelines for response to intraoperative neuromonitoring events in high-risk spinal deformity surgery. Spine Deform. 2022;10:745-761.
- Welling SE, Bauer JM. Delayed postoperative spinal cord ischemia after posterior spinal fusion in a pediatric patient with syrinx and decompressed Chiari: a case report. JBJS Case Connect. 2020;10(3):e1900610.
- Kia C, Stelzer JW, Lee MC. Delayed postoperative spinal cord injury with complete paralysis after adolescent idiopathic surgery: a case report. JBJS Case Connect. 2022;12(1). doi: 10.2106/JBJS.CC.21.00497.
- Quinonez A, Pahys JM, Samdani AF, et al. Complete paraplegia 36 h after attempted posterior spinal fusion for severe adolescent idiopathic scoliosis: a case report. Spinal Cord Ser Cases. 2021;7(1):33.
- Lovi A, Manfroni F, Luca A, et al. Delayed postoperative cervical spinal cord ischemic lesion after a thoracolumbar fusion for syndromic scoliosis: a case report and systematic review of the literature. Childs Nerv Syst. 2022;38(5):1011-1015.
- Chang JH, Hoernschemeyer DG, Sponseller PD. Delayed postoperative paralysis in adolescent idiopathic scoliosis: management with partial removal of hardware and staged correction. J Spinal Disord Tech. 2006;19(3):222-225.
- Alam M, Shufflebarger HL, Rush AJ, et al. Delayed quadriparesis after posterior spinal fusion for scoliosis: a case series. Spine Deform. 2020;8(5):1075-1080.
- Lewis ND, Keshen SG, Lenke LG, et al. The deformity angular ratio: does it correlate with high-risk cases for potential spinal cord monitoring alerts in pediatric 3-column thoracic spinal deformity corrective surgery? Spine (Phila Pa 1976). 2015;40(15):E879-E885.
- Wang XB, Lenke LG, Thuet E, et al. Deformity angular ratio describes the severity of spinal deformity and predicts the risk of neurologic deficit in posterior vertebral column resection surgery. Spine (Phila Pa 1976). 2016;41(18):1447-1455.
- Sielatycki JA, Cerpa M, Baum G, et al. A novel MRI-based classification of spinal cord shape and CSF presence at the curve apex to assess risk of intraoperative neuromonitoring data loss with thoracic spinal deformity correction. Spine Deform. 2020;8(4):655-661.
- Cheh G, Lenke LG, Padberg AM, et al. Loss of spinal cord monitoring signals in children during thoracic kyphosis correction with spinal osteotomy: why does it occur and what should you do? Spine (Phila Pa 1976). 2008;33(10):1093-1099.
- Coe JD, Smith JS, Berven S, et al. Complications of spinal fusion for scheuermann kyphosis: a report of the scoliosis research society morbidity and mortality committee. Spine (Phila Pa 1976). 2010;35(1):99-103.
- Kamerlink JR, Errico T, Xavier S, et al. Major intraoperative neurologic monitoring deficits in consecutive pediatric and adult spinal deformity patients at one institution. Spine (Phila Pa 1976). 2010;35(2): 240-245.
- Burton DC, Carlson BB, Place HM, et al. Results of the Scoliosis Research Society Morbidity and Mortality Database 2009-2012: A Report From the Morbidity and Mortality Committee. Spine Deform. 2016;4(5):338-343.
- Roye BD, Campbell ML, Matsumoto H, et al. Establishing consensus on the best practice guidelines for use of halo gravity traction for pediatric spinal deformity. J Pediatr Orthop. 2020;40(1):e42-e48.
- Buchowski JM, Bhatnagar R, Skaggs DL, et al. Temporary internal distraction as an aid to correction of severe scoliosis. J Bone Joint Surg Am. 2006;88(9):2035-2041.
- Horn SR, Poorman GW, Tishelman JC, et al. Trends in treatment of Scheuermann kyphosis: a study of 1,070 cases from 2003 to 2012. Spine Deform. 2019;7(1):100-106.
- Floccari LV, Poppino K, Greenhill DA, et al. Ponte osteotomies in a matched series of large AIS curves increase surgical risk without improving outcomes. Spine Deform. 2021;9(5):1411-1418.
- Samdani AF, Bennett JT, Singla AR, et al. Do ponte osteotomies enhance correction in adolescent idiopathic scoliosis? An analysis of 191 Lenke 1A and 1B curves. Spine Deform. 2015;3(5):483-488.
- Singla A, Bennett JT, Sponseller PD, et al. Results of selective thoracic versus nonselective fusion in Lenke type 3 curves. Spine (Phila Pa 1976). 2014;39(24):2034-2041.
- McCann ME, Brustowicz RM, Bacsik J, et al. The bispectral index and explicit recall during the intraoperative wake-up test for scoliosis surgery. Anesth Analg. 2002;94(6):1474-1478.
- Altaf F, Griesdale DE, Belanger L, et al. The differential effects of norepinephrine and dopamine on cerebrospinal fluid pressure and spinal cord perfusion pressure after acute human spinal cord injury. Spinal Cord. 2017;55(1):33-38.
- Backster A, Teo A, Swift M, et al. Transforming the surgical “time-out” into a comprehensive “preparatory pause”. J Card Surg 2007;22(5):410-416.
- Verma K, Lonner B, Dean L, et al. Reduction of mean arterial pressure at incision reduces operative blood loss in adolescent idiopathic scoliosis. Spine Deform. 2013;1(2):115-122.
- Andras L, Louie K, Skaggs DL. Weak or absent ankle dorsiflexion: the most sensitive indicator of motor deficits following spinal deformity surgery. Spine Deform. 2014;2(3):198-202.
- Haber LL, Womack ED, Sathyamoorthy M, et al. Who needs a pediatric intensive care unit after posterior spinal fusion for adolescent idiopathic scoliosis? Spine Deform. 2018;6(2):137-140.
- Shan LQ, Skaggs DL, Lee C, et al. Intensive care unit versus hospital floor: a comparative study of postoperative management of patients with adolescent idiopathic scoliosis. J Bone Joint Surg Am. 2013;95(7):e40.
- Fletcher ND, Andras LM, Lazarus DE, et al. Use of a novel pathway for early discharge was associated with a 48% shorter length of stay after posterior spinal fusion for adolescent idiopathic scoliosis. J Pediatr Orthop. 2017;37(2):92-97.
- Fletcher ND, Murphy JS, Austin TM, et al. Short term outcomes of an enhanced recovery after surgery (ERAS) pathway versus a traditional discharge pathway after posterior spinal fusion for adolescent idiopathic scoliosis. Spine Deform. 2021;9(4):1013-1019.
- Fletcher ND, Shourbaji N, Mitchell PM, et al. Clinical and economic implications of early discharge following posterior spinal fusion for adolescent idiopathic scoliosis. J Child Orthop. 2014;8(3):257-263.
- Bellaire LL, Bruce RW Jr, Ward LA, et al. Use of an accelerated discharge pathway in patients with severe cerebral palsy undergoing posterior spinal fusion for neuromuscular scoliosis. Spine Deform. 2019;7(5):804-811.
- Fletcher ND, Bellaire LL, Dilbone ES, et al. Variability in length of stay following neuromuscular spinal fusion. Spine Deform. 2020;8(4):725-732.
- Hwang JY, Sohn HM, Kim JH, et al. Reproducible motor deficit following aortic occlusion in a rat model of spinal cord ischemia. J Vis Exp. 2017;(125):e55814. doi: 10.3791/55814.
