Current Concept Review
Nerve Transfers in the Lower Extremity
1Boston Children’s Hospital, Department of Orthopedics, Harvard Medical School, Boston, MA; 2Northwestern Memorial Hospital, Department of Orthopaedic Surgery, Feinberg School of Medicine, Chicago, IL
Correspondence: Andrea S. Bauer, MD, Boston Children’s Hospital, 300 Longwood Ave., Boston, MA 02115. E-mail: [email protected]
Received: October 18, 2022; Accepted: December 14, 2022; Published: February 1, 2023
Volume 5, Number 1, February 2023
Abstract
The utilization of nerve transfer procedures in the upper extremity following brachial plexus injury, trauma, spinal cord injury, tumors, infection, or other etiologies are well established. Nerve injuries in the lower extremity pose several additional challenges, including longer distance to target motor end plates, delayed presentation, and concomitant limb trauma. Nerve transfers in the lower extremity have the potential to provide functional (sensory or motor) recovery distally after direct surgical coaptation of a functional donor nerve to a non-functional recipient nerve. The ability to perform pure motor or sensory fascicular transfers allows for focused recovery while limiting donor morbidity. Indications for nerve transfers in the lower extremity are evolving, but transfers have been utilized for non-recovering peroneal, obturator, femoral, or tibial nerve palsies, to provide protective sensation to the plantar aspect of the foot as well as for painful neuropathies/neuromas. There is a paucity of orthopaedic literature on this topic and our review aims to highlight the current state of lower extremity nerve transfers as they relate to the practicing orthopaedist, including future directions in the field.
Key Concepts
- Nerve transfers have been well-established as a treatment option for nerve injuries of the upper extremity and brachial plexus, but their use in the lower extremities is less common.
- Nerve transfers may be of particular interest in the lower extremity because of the ability to cover relatively long distances as well as avoid the zone of injury, especially in far proximal injuries.
- Nerve transfers of the lower extremity have been used to address motor deficit of the peroneal, femoral, obturator, and tibial nerves, as well as tibial and sural nerve sensory loss.
- There is currently a paucity of orthopaedic literature on lower extremity nerve transfers and further understanding is required to better utilize these techniques to manage lower extremity peripheral nerve injury.
Introduction
The utilization of nerve transfer procedures in the upper extremity following brachial plexus injury, trauma, spinal cord injury, tumors, infection, or other etiologies is well established.1–3 Less common, however, are nerve transfer procedures of the lower extremity. Currently, investigations into nerve transfer of the lower extremity are limited in the literature, even though nerve injury can be a common occurrence encountered by orthopaedists after trauma, complications following total joint arthroplasty, or oncologic surgery.4,5 This review details the existing literature on lower extremity nerve transfers with a focus on orthopaedic applications, including future directions in the field.
Peripheral nerve injury (PNI) has been classified by the Sunderland classification as neurapraxia, axonotmesis, or neurotmesis.6,7 Prior research has shown that PNI has been found in 1.8% of lower extremity trauma and that those with PNI are more likely to be younger men involved in motorcycle crashes or to have a penetrating injury.5 A thorough exam at the time of injury helps to establish a baseline for treatment. Motor function of peripheral nerves is graded using the British Medical Research Council (BMRC) muscle grading system. A score of 0 indicates no muscle contraction, while a 1 is a flicker of muscle activity, 2 is full motion with gravity eliminated, 3 is anti-gravity strength, 4 is less than full and 5 is full motor strength.8 If there is a nerve injury, observation with electromyography (EMG) by 3 months has been advocated as an initial strategy to evaluate for a neurapraxia. Nerve injuries that do not resolve by this time suggest either more complete injury or continued insult such as nerve compression. Advanced imaging such as MRI can help delineate the location of a PNI while surgical exploration can free compressive injuries as well as evaluate for the potential of primary nerve repair.7
Primary repair is indicated if the nerve is sharply sectioned without a wide zone of injury and that can be brought together tension-free. For injuries that cannot be repaired tension-free, autogenous nerve graft such as the sural nerve has been the gold standard of repair but requires intact proximal and distal nerve targets with a donor nerve with function that either can be sacrificed or is redundant.9 Nerve conduits can be used to direct nerve regeneration, although their use is typically reserved for shorter segment defects. If direct repair or graft is not possible, nerve transfer with direct surgical coaptation of a functional donor nerve to a non-functional recipient nerve can be used.
Nerve transfers are of interest in lower extremity PNI given the long distance that nerve grafts may have to cover as well as the ability to avoid the zone of injury, especially in far-proximal injuries. In a study of peroneal nerve injuries, Kim et al. found that 88% of common peroneal nerve stretch/compression injuries treated with neurolysis regained useful function while 84% of patients requiring direct repair regained useful function. When nerve graft was required, likelihood of recovery was inverse to the length of graft required, with 75% of patients requiring graft repair of under 6 cm in length recovering at least M3 peroneal function, while 38% of patients with grafts of 6-12 cm recovered useful peroneal function and only 16% of patients with graft length over 12 cm recovered useful function.10
Recovery following any nerve repair is time sensitive, as nerve regeneration occurs at a rate of 1 mm/day and distal muscle groups may fibrose and no longer be candidates for reinnervation after a period of 12–18 months.7 Nerve transfer taken from the most distal segment of the donor nerve coapted to the most proximal viable segment of the recipient nerve shortens the distance, and therefore time, for nerve regeneration to occur. While there is the possibility of donor site morbidity, preservation of function can be expected even with up to 80% of the total axons of a nerve harvested.11,12
In purely sensory deficits or in partial motor deficit, a “Supercharged End to Side” transfer (SETS) has been proposed to preserve and possibly augment recovering function distal to the zone of injury. Peters et al. in their work on femoral nerve palsies using either sartorius, adductor longus or gracilis donor motor branches demonstrated recovery from a mean preoperative MRC score of 2.33 to at least MRC grade 4+ following SETS transfer for partial femoral nerve palsy. In this study, patients with motor unit action potentials (MUAP) at the recipient nerve as well as intraoperative response to electrical stimulation underwent SETS alone, while patients with either present MUAP or intraoperative response to stimulation received both SETS and end to end transfer (ETE). Patients with no response underwent ETE alone.12
Indications and Contraindications
Nerve transfer is indicated when PNI is identified with the following: 1) Absence of any motor function at 3 to 12 months after injury; 2) Gap between nerve ends too long for primary repair or grafting; and 3) Proximal nerve end not available for repair. The goals of recovery is a stable hip joint capable of bearing weight, which utilizes the hip abductor complex. Next is the ability to ambulate with a normal gait, focusing on the quadriceps and hamstrings proximally as well as push-off and ankle dorsiflexion distally. Protective sensation of the plantar foot is also of importance, preventing the formation of ulcers that can cause infection.13 To this end, nerve transfers in the lower extremity have been utilized for non-recovering peroneal, obturator, femoral, or tibial nerve palsies to provide protective sensation to the plantar aspect of the of foot as well as for painful neuropathies/neuromas.14–17
In general, nerve transfers are not the preferred surgical technique when other surgical options will provide similar or improved outcomes with less morbidity, and treatment should follow the least invasive or most likely to succeed step from the reconstructive ladder above. Contraindications for motor nerve transfers include delay greater than 12-18 months since time of injury or donor nerve strength is less than BMRC grade M4.3
General Principles
Motor Nerve Transfers: Motor nerve transfers require direct end-to-end coaptation. The recipient nerve should first be dissected and directly stimulated to confirm the absence of muscle contraction. If there is present motor activity, neurolysis at any site of compression should be done and SETS transfer can be considered. Once absence of motor function is confirmed, the donor nerve is also dissected and its function assessed by direct electrical stimulation. This helps to confirm the specific motor function of an individual fascicle transfer in order to selectively sacrifice a nonessential motor function (e.g., toe flexors of the tibial nerve to peroneal branch to tibialis anterior). Additionally, redundant innervation of a given muscle can be identified, allowing for harvest of similar axon numbers from donor to recipient as is the case when harvesting multiple branches of the anterior obturator nerve to the femoral nerve. This is of importance because axonal mismatch has been hypothesized as a cause of failure in nerve transfers, and as a consequence, the relative size of the nerves planned for transfer must be considered preoperatively.9,11 If individual fascicular transfer is being performed, internal neurolysis of the donor nerve is performed and function of the donor fascicle and the remaining donor nerve is assessed.3 Once the appropriate branches or fascicles are selected, coaptation follows the mantra of “donor distal, recipient proximal” to ensure enough length of each fascicle for a tensionless repair.18 The recipient nerve must be carefully evaluated to determine the zone of injury to assess the proximal most point where coaptation to healthy nerve tissue is possible. The donor nerve is cut sharply distally to achieve the necessary length for grafting. The coaptation can then be performed using microsurgical technique in the standard fashion with a tension-free repair. Importantly, sites of compression along the transfer must be identified and freed as necessary to prevent any impediment to recovery.12
Sensory Nerve Transfers: Similar to motor nerve transfers, end-to-end coaptation is preferred in sensory nerve transfers; however, end-to-side transfers are also successful in providing protective sensation.3 General principles of nerve transfer are otherwise the same between sensory and motor nerve transfers. In sensory transfers, a donor nerve that does not provide critical sensation is used in order to regain critical sensation once provided by the recipient nerve. Direct stimulation of the target nerve can help prevent unintentional motor nerve harvest. Sensory nerve transfers of the lower extremity have been utilized to provide protective sensation to the plantar aspect of the foot as well as for painful neuromas of the deep peroneal nerve (DPN).14,17,19
Nerve Transfers for Peroneal Nerve Dysfuntion
The peroneal nerve is the most commonly injured nerve in lower extremity trauma, representing an estimated 50% of traumatic lower extremity nerve injuries.5 Peroneal nerve injury can also be iatrogenic (e.g., prolonged stretching, direct compression or trauma during surgical procedures) or from a space-occupying malignancy causing compression of the nerve. Physical exam will demonstrate a foot drop with loss of ankle dorsiflexion and eversion as well as toe extension secondary to loss of the tibialis anterior, extensor hallicus longus, and peroneal muscles as well as diminished sensation over the anterolateral lower leg and dorsum of the foot. Treatment typically begins with fitting of a custom ankle foot orthosis (AFO) to accommodate for the foot drop along with physical therapy and observation of the nerve recovery. If no recovery is seen by 2-4 months of observation alone, surgical intervention can be considered.7,10,12,20 Surgical options follow the treatment algorithm outlined above. Tendon transfer is the most common surgical treatment, although it may not restore normal gait, and long-term development of worsening deformity and arthritis in the foot and ankle is possible.21
Several cadaveric and in vivo studies have examined the feasibility of using proximal motor branches of the tibial nerve as donors for common peroneal nerve (CPN) or deep peroneal nerve (DPN) injuries.15,21–23 The first series describing motor nerve transfers for peroneal nerve injuries was in an eight-patient cohort of pediatric patients in Iran who underwent diclofenac injection in the gluteal region resulting in common peroneal nerve palsy and one patient who had transection of the CPN after a stabbing injury.24 The treating surgeons performed nerve transfers from the soleus and lateral gastrocnemius branches of the tibial nerve to the DPN in an attempt to regain ankle dorsiflexion. Of the nine patients, seven had at least M4 recovery of ankle dorsiflexion by 6 months, M4 recovery of extensor hallucis longus (EHL) by 8 months, and M3-4 recovery of remaining toe extension by 10 months.24 The two patients who did not have functional recovery had a CPN palsy prior to nerve transfer of 14 and 18 months, which is likely beyond the timeframe in which neuromuscular end plates remain viable.7
Results have been inconsistent, however, with several studies showing less than 50% of patients recovering significant muscle strength by final follow-up. Leclere et al. performed isolated partial tibial nerve to CPN transfers on six patients with CPN palsies and had mixed results, with only 3/6 (50%) patients achieving M4 ankle dorsiflexion scores at final follow-up.23 Giuffre et al. similarly performed partial tibial to CPN transfers on 11 patients with CPN palsies due to various etiologies and reported MRC grades of 3/5 or greater in only 4/11 (36.4%) patients.21 Flores et al. performed transfers of the tibial motor nerve to the soleus to the deep peroneal nerve, with only three out of 10 (30%) patients regaining BMRC grades of 3 or higher.16 However, Nath et al. reported BMRC grade of 3 or higher in 11/14 (78.5%) of patients with deep peroneal nerve injuries treated with nerve transfer from either the tibial or superficial peroneal nerve.25 One possible explanation for this inconsistency is suggested by Crowe et al., who found that failure after partial tibial nerve transfer to the deep peroneal nerve was associated with mismatch of donor axon quantity to recipient axon size.11 However, nerve transfer remains a viable option in the appropriately selected patient, and those who fail to demonstrate adequate recovery may still be candidates for tendon transfer.
Fascicles of the tibial nerve innervating the flexor digitorum longus (FDL) or flexor hallicus longus (FHL) have been chosen as donor nerves for transfer given their redundant innervation, the presence of stronger push-off from gastrocnemius and soleus muscles, as well as the possibility for compensation by remaining intrinsic foot musculature.11 Their transfer to the DPN is typically performed with the patient in a lateral position. An oblique incision is made centered over the fibular head. The CPN is identified and visualized as it branches into the DPN and SPN. After decompression of the DPN, electrical stimulation is used to confirm lack of motor function in the tibialis anterior or toe extensors. The interval between the peroneus longus and soleus is opened, and the peroneus longus is elevated off the anterior fibula to visualize the DPN branches into the tibialis anterior muscle. The branching pattern is variable here, so direct visualization of the nerve branches into the tibialis anterior is essential. Similarly, the soleus is elevated off the posterior fibula to identify the posterior tibial neurovascular bundle. Intra-fascicular dissection of the tibial nerve is performed to identify a redundant fascicle to the FDL or FHL. The recipient nerve is transected proximally and the donor nerve is transected distally to give sufficient length for transfer. If needed, the tibial fascicle can be passed through the interosseous membrane for additional length; however, meticulous care must be taken to ensure there is no site of compression along the transfer. Tension-free coaptation is performed using multiple interrupted 9.0 nylon epineurial sutures along with fibrin glue (Video 1).
Other Motor Nerve Transfers
Femoral, obturator, and tibial nerve palsies can also occur due to trauma, direct compression, maternal obstetric complications or iatrogenic injury from intra-pelvic surgery, or total joint arthroplasty. These can be devastating injuries that severely hinder patients’ ability to ambulate with a normal gait, even with assistive devices. Table 1 summarizes the lower extremity motor nerve transfers that have been described to date.
Table 1. Lower Extremity Motor Nerve Transfers
| Etiology | Motor Deficit | Donor Nerve | Recipient Nerve |
|---|---|---|---|
| Peroneal Nerve Injury (CPN/DPN) - Traumatic - Iatrogenic - Tumor |
Ankle Dorsiflexion, eversion | Proximal motor branches of tibial nerve (Medial/Lateral Gastrocnemius branches, posterior branch to soleus, branches to FHL/FDL) | Common Peroneal Nerve (CPN) or Deep Peroneal Nerve (DPN) |
| Femoral Nerve Palsy | Knee Extension | Anterior branch of obturator nerve, Sartorius nerve branches | Femoral Nerve |
| Obturator Nerve Palsy - Obstetric - Iatrogenic |
Thigh Adduction | Proximal branch of femoral nerve | Obturator Nerve |
| Tibial Nerve Palsy | Ankle Plantarflexion | Femoral Nerve branches or anterior branch of obturator nerve | Medial/Lateral Gastrocnemius branches of tibial nerve |
Femoral nerve palsies affecting the quadriceps musculature can both weaken stabilization of the knee in stance as well as prevent normal knee extension during gait. Anatomic studies have demonstrated the feasibility of motor branches of the obturator nerve to femoral nerve transfers in the case of femoral nerve palsies.26,27 Regaining femoral nerve function allows for adequate knee extension, and studies have shown that using branches of the anterior obturator nerve or nerves to the sartorius can restore near-normal gait without significant loss of adductor function.12,28–32 Figure 1 depicts the transfer of the gracilis branch of the obturator nerve to the rectus femoris branch of the femoral nerve in a patient with a femoral nerve laceration and large zone of injury.
Figure 1. Clinical photographs of gracilis motor branch to rectus femoris motor branch transfer. A) Long oblique incision marked on skin to include both exploration of the femoral nerve and planned nerve transfer. B) Lacerated branches of the femoral nerve (yellow arrows). C) The gracilis motor branch of the obturator nerve (black arrow) is passed deep to the adductor muscles and superficial to the femoral vessels to reach the rectus femoris motor branch (yellow arrow) of the femoral nerve. D) View through the microscope after coaptation of the nerves using microsutures and fibrin glue.
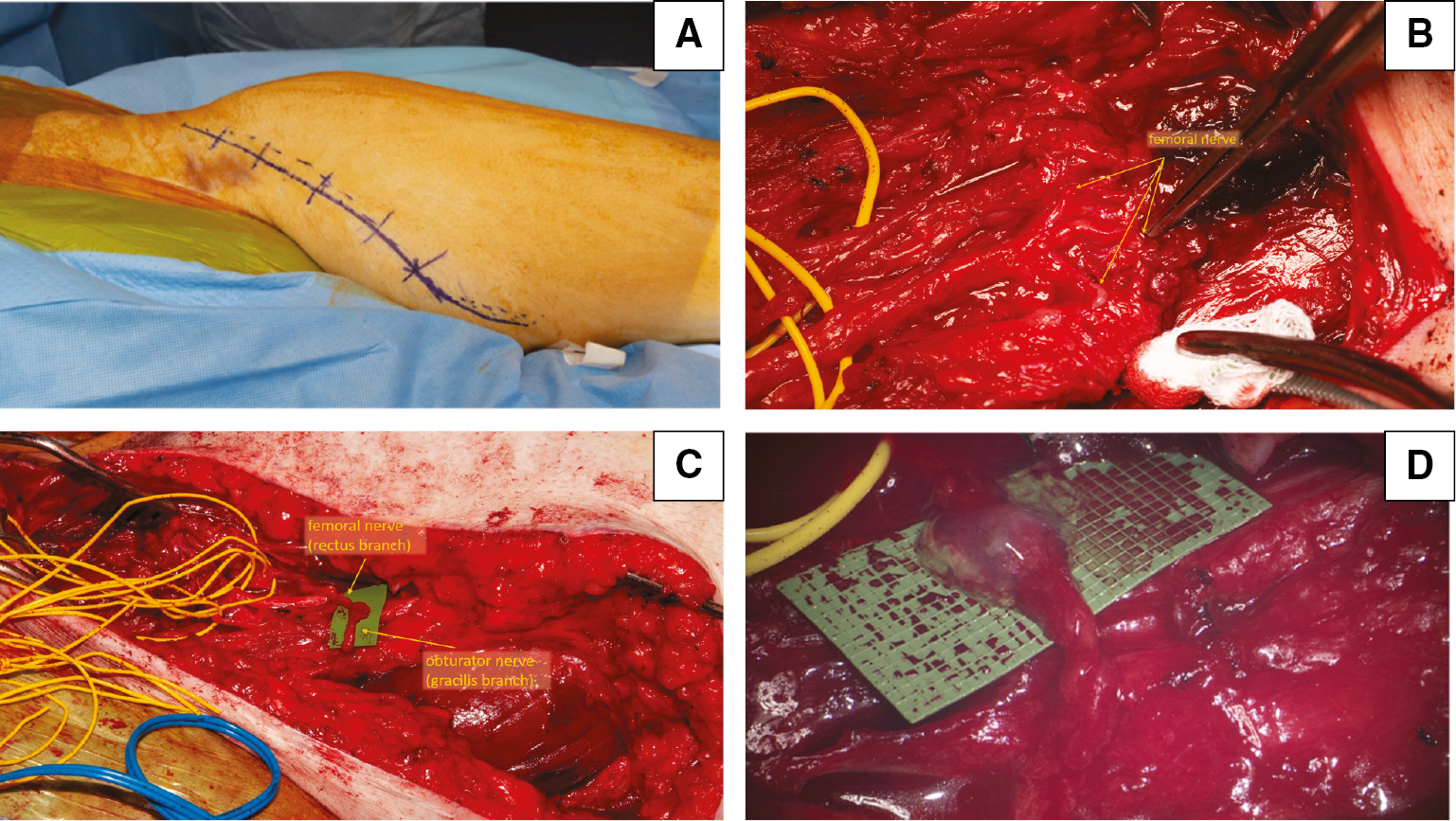
Additionally, Spilopoulos et al. demonstrated the effectiveness of nerve transfer in the reverse, using a proximal branch of the femoral nerve to obturator nerve for hip adduction deficits in a patient after pelvic surgery for gynecologic malignancy.33
Tibial nerve function to the gastrocnemius and soleus muscles is crucial in providing push-off power for a normal gait. Prior cadaveric studies investigating tibial nerve palsies have demonstrated the potential to use distal femoral nerve branches to vastus medialis and vastus lateralis as donor nerves to the medial and lateral gastrocnemius branches of the tibial nerve.28,29 Work by Moore and Yin has demonstrated return of function in ankle plantarflexion after nerve transfers from the femoral and anterior branch of obturator nerve to the gastrocnemius branches of the tibial nerve.29,34
Sensory Nerve Transfers
Maintaining protective sensation over the plantar aspect of the foot is of paramount importance as loss of plantar sensation can lead to ulcer formation and infection with resulting amputation.19 Sensory nerve transfers to the tibial nerve from the saphenous, sural, or branches of superficial or deep peroneal nerve offer the ability to restore sensibility to the plantar foot.17 Table 2 lists the sensory nerve transfers that have been described for the lower extremity.
Table 2. Lower Extremity Sensory Nerve Transfers
| Etiology | Sensory Deficit | Donor Nerve | Recipient Nerve |
|---|---|---|---|
| Tibial Nerve Injury | Lack of protective plantar sensation of the foot | Saphenous Nerve/Sural Nerve Or Distal aspect of DPN |
Tibial Nerve Or Medial plantar branch of tibial nerve |
| Sural Nerve Injury | Lateral foot/ankle numbness | Saphenous Nerve | Sural Nerve |
| Neuroma Formation - Amputation - Iatrogenic |
Painful Neuroma (in corresponding peripheral nerve distribution) | Tibial Nerve DPN Symptomatic neuroma following amputation (TMR) |
CPN Superficial Peroneal Nerve (SPN) Functional motor nerve in adjacent musculature (TMR) |
Koshima et al. describe a technique by which the distal-most aspect of the DPN is identified in the first web space and subcutaneously tunneled to the medial plantar branch of the posterior tibial nerve with successful return of protective sensation in that distribution.17 A study by Agarwal et al. demonstrated not only restored sensation but improved healing of chronic plantar ulcers present in patients with leprosy after saphenous to posterior tibial nerve transfer.19 Importantly, when compared to motor transfers, the sensory saphenous nerve transfers in this study were not limited by loss of motor end plates after 12-18 months and in fact, sensibility was able to be restored after up to 5 years of complete anesthesia at the plantar aspect of the foot.
The management of painful neuromas can be challenging, and multiple surgical and non-surgical treatment methods exist ranging from gabapentin, neuroma ablation or excision, traction neurectomy, or burying the neuroma in adjacent structures.35 Nerve transfers have demonstrated a promising treatment option for this difficult problem. Economides et al. demonstrated the effectiveness of tibial to peroneal nerve coaptation at the time of trans-femoral amputation to decrease painful neuroma formation and phantom leg pain. At 6 months, patients who underwent coaptation had improved visual analog scale (VAS) scores, less phantom leg pain, and were over seven times as likely to be ambulating with a prosthesis when compared to patients who underwent standard traction neurectomy.36 Bibbo et al. demonstrated improved VAS scores in patients with SPN neuromas following anterior approach to the ankle for arthrodesis who were treated with SPN to DPN transfers combined with intermediary nerve allograft.14 Perhaps the most exciting recent development is that of targeted muscle reinnervation (TMR). Originally developed with an aim of powering myoelectric prosthesis, this technique has emerged as a viable option to decrease pain and improve function following neuroma formation. In this technique, the neuroma is excised to a healthy base of nerve fascicles which are coapted to a motor nerve innervating a nearby muscle. This has been shown to reduce both phantom pain and residual limb pain as well as improved PROMIS, Neuro-QOL, and OPUS Rasch scores in major limb amputees.37 Research by Chang et al. into highly comorbid below knee amputation patients also found that patients with TMR were not only significantly likely to have less phantom limb pain or residual limb pain, they were also more likely than patients with traction neurectomy to be ambulatory and off opiate mediation.35
Future Directions–Motor Transfers for Acute Flaccid Myelitis
Acute Flaccid Myelitis (AFM) is an acute neurologic process that affects the grey matter of the spinal cord in children, preferentially affecting the anterior horn cells resulting in a ‘polio-like’ presentation. Standard of care consists of corticosteroids and intravenous immunoglobulins, though recovery of motor function is highly variable and permanent loss of function is possible.38 Nerve transfers have emerged as a promising option for patients with permanent motor functional loss due to AFM, following similar principles to those for other indications. Moore et al. outlined a case series of eight patients with AFM, with transfers for gluteal, femoral, hamstring and gastrocnemius function performed.13 Additionally, Lubelski et al. demonstrated sciatic to femoral nerve transfer for obturator and quadriceps paresis in four patients with AFM.32 Doi et al. additionally reported the use of contralateral obturator to restore left femoral nerve function 14 months postoperatively.39 Nerve transfers are a potentially powerful tool in the treatment of AFM, as typically grafting of these proximal lesions is not possible.
Complications
The studies evaluated for this review did not find significant rates of major operative complications such as hemorrhage or infection. Historically, there has been concern for donor site nerve morbidity; however, judicious selection of either redundant innervation or functionally superfluous muscles can avoid this complication. Additionally, leaving at least 20% of the axons to a given muscle remain following harvest has been reported to result in normal motor function.11,12 Mild sensory deficits have been found in patients with obturator transfer to the femoral nerve, but patients did not find this sensory loss to be limiting.40 Deficits after branches of the tibial nerve are used as donor nerves include deficits in toe flexion, decreased calf circumference, or decreased ankle plantarflexion.25 However, these deficits must be weighed against the potential benefits of a successful nerve transfer.
Summary
While nerve transfers have long been used in the upper extremity for both motor and sensory deficits, their utility in the lower extremities is relatively new. Nerve transfers offer the potential to restore function to peripheral nerve injuries requiring large distances of nerve autograft or in those injuries without sufficient or accessible proximal nerve endings for grafting. Appropriate selection of donor nerve can minimize donor site loss of function by utilizing nerves with redundant innervation or those powering muscles with redundant or superfluous function. Taken in total, nerve transfers offer an important step in the reconstructive ladder for patients with peripheral nerve injury in the lower extremity as can commonly be seen by the practicing orthopaedist. Given the promising outcomes of lower extremity nerve transfers currently in the literature, it is likely their use will continue to expand as the orthopedic community becomes more familiar with potential indications.
Additional Link
- American Academy of Orthopaedic Surgeons (AAOS) Orthopaedic Video Theatre, Surgical Management of Complete Footdrop via Partial Tibial Nerve Transfer to the Motor Branch of the Tibialis Anterior.
Disclaimer
No funding was received. The authors have no conflicts of interest to report related to the subject matter.
References
- Giuffre JL, Kakar S, Bishop AT, et al. Current concepts of the treatment of adult brachial plexus injuries. J Hand Surg Am. 2010;35(4):678–688.
- Kozin SH. Nerve transfers in brachial plexus birth palsies: indications, techniques, and outcomes. Hand Clin. 2008;24(4):363-376.
- Lee SK, Wolfe SW. Nerve transfers for the upper extremity: new horizons in nerve reconstruction. J Am Acad Orthop Surg. 2012;20(8):506-517.
- Hasija R, Kelly JJ, Shah NV, et al. Nerve injuries associated with total hip arthroplasty. J Clin Orthop Trauma. 2018;9(1):81-86.
- Huckhagel T, Nüchtern J, Regelsberger J, et al. Nerve trauma of the lower extremity: evaluation of 60,422 leg injured patients from the TraumaRegister DGU® between 2002 and 2015. Scand J Trauma Resusc Emerg Med. 2018;26(1):1-8.
- Sunderland S. A classification of peripheral nerve injuries producing loss of function. Brain. 1951;74(4):491-516.
- Griffin JW, Hogan MCV, Chhabra AB, et al. Peripheral nerve repair and reconstruction. J Bone Jt Surg - Ser A. 2013;95(23):2144-2151.
- Wang Y, Sunitha M, Chung KC. How to measure outcomes of peripheral nerve surgery. Hand Clin. 2013;29(3):349-361.
- Ray WZ, Mackinnon SE. Nerve problems in the lower extremity. Foot Ankle Clin. 2011;16(2):243-254.
- Kim DH, Murovic JA, Tiel RL, et al. Management and outcomes in 318 operative common peroneal nerve lesions at the Louisiana State University Health Sciences Center. Neurosurgery. 2004;54(6):1421-1429.
- Crowe CS, Mosca VS, Osorio MB, et al. Partial tibial nerve transfer for foot drop from deep peroneal palsy: lessons from three pediatric cases. Microsurgery. 2022;42:71-75.
- Peters BR, Ha AY, Moore AM, et al. Nerve transfers for femoral nerve palsy: an updated approach and surgical technique. J Neurosurg. 2021;136:856-866.
- Moore AM, Bettlach CR, Tung TT, et al. Lower extremity nerve transfers in acute flaccid myelitis patients: a case series. Plast Reconstr Surg Glob Open. 2021;9:e3699.
- Bibbo C, Rodrigues-Colazzo E, Finzen AG. Superficial peroneal nerve to deep peroneal nerve transfer with allograft conduit for neuroma in continuity. J Foot Ankle Surg. 2018;57(3):514-517.
- Strazar R, White CP, Bain J. Foot reanimation via nerve transfer to the peroneal nerve using the nerve branch to the lateral gastrocnemius: case report. J Plast Reconstr Aesthetic Surg. 2011;64(10):1380-1382.
- Flores LP, Martins RS, Siqueira MG. Clinical results of transferring a motor branch of the tibial nerve to the deep peroneal nerve for treatment of foot drop. Neurosurgery. 2013;73(4):609-615.
- Koshima I, Nanba Y, Tsutsui T, et al. Deep peroneal nerve transfer for established plantar sensory loss. J Reconstr Microsurg. 2003;19(7):451-454.
- Tung TH, Mackinnon SE. Nerve transfers: indications, techniques, and outcomes. J Hand Surg Am. 2010;35(2):332-341.
- Agarwal P, Shukla P, Sharma D. Saphenous nerve transfer: a new approach to restore sensation of the sole. J Plast Reconstr Aesthetic Surg. 2018;71(12):1704-1710.
- Mont MA, Dellon AL, Chen F, et al. The operative treatment of peroneal nerve palsy. J Bone Jt Surg - Ser A. 1996;78(6):863-869.
- Giuffre JL, Bishop AT, Spinner RJ, et al. Partial tibial nerve transfer to the tibialis anterior motor branch to treat peroneal nerve injury after knee trauma. Clin Orthop Relat Res. 2012;470(3):779-790.
- Bodily KD, Spinner RJ, Bishop AT. Restoration of motor function of the deep fibular (Peroneal) nerve by direct nerve transfer of branches from the tibial nerve: an anatomical study. Clin Anat. 2004;17(3):201-205.
- Leclère FM, Badur N, Mathys L, et al. Nerve transfers for persistent traumatic peroneal nerve palsy: the inselspital bern experience. Neurosurgery. 2015;77(4):572-579.
- Gousheh J, Babaei A. A new surgical technique for the treatment of high common peroneal nerve palsy. Plast Reconstr Surg. 2002;109(3):994-998.
- Nath RK, Lyons AB, Paizi M. Successful management of foot drop by nerve transfers to the deep peroneal nerve. J Reconstr Microsurg. 2008;24(6):419-427.
- Goubier J-N, Teboul F, Yeo S. Transfer of two motor branches of the anterior obturator nerve to the motor portion of the femoral nerve: an anatomical feasibility study. Microsurgery. 2012;32(6):463-465.
- Tung TH, Chao A, Moore AM. Obturator nerve transfer for femoral nerve reconstruction: anatomic study and clinical application. Plast Reconstr Surg. 2012;130(5):1066-1074.
- Meng D, Chen H, Lin Y, et al. Transferring of femoral nerve motor branches for high-level sciatic nerve injury: a cadaver feasibility study. Acta Neurochir (Wien). 2019;161(2):279-286.
- Moore AM, Krauss EM, Parikh RP, et al. Femoral nerve transfers for restoring tibial nerve function: an anatomical study and clinical correlation: a report of 2 cases. J Neurosurg. 2018;129(4):1024-1033.
- Campbell AA, Eckhauser FE, Belzberg A, et al. Obturator nerve transfer as an option for femoral nerve repair: case report. Neurosurgery. 2010;66(6 Suppl Operative):375.
- Rastrelli M, Tocco-Tussardi I, Tropea S, et al. Transfer of the anterior branch of the obturator nerve for femoral nerve reconstruction and preservation of motor function: a case report. Int J Surg Case Rep. 2018;51:58-61.
- Lubelski D, Pennington Z, Tuffaha S, et al. Sciatic-to-femoral nerve end-to-end coaptation for proximal lower extremity function in patients with acute flaccid myelitis: technical note and review of the literature. Oper Neurosurg. 2021;21(1):20-26.
- Spiliopoulos K, Williams Z. Femoral branch to obturator nerve transfer for restoration of thigh adduction following iatrogenic injury: case report. J Neurosurg. 2011;114(6):1529-1533.
- Yin G, Chen H, Hou C, et al. Obturator nerve transfer to the branch of the tibial nerve innervating the gastrocnemius muscle for the treatment of sacral plexus nerve injury. Neurosurgery. 2016;78(4):546-551.
- Chang BL, Mondshine J, Attinger CE, et al. Targeted muscle reinnervation improves pain and ambulation outcomes in highly comorbid amputees. Plast Reconstr Surg. 2021;148:376-386.
- Economides JM, Defazio MV, Attinger CE, et al. Prevention of painful neuroma and phantom limb pain after transfemoral amputations through concomitant nerve coaptation and collagen nerve wrapping. Neurosurgery. 2016;79(3):508-512.
- Mioton LM, Dumanian GA, Shah N, et al. Targeted muscle reinnervation improves residual limb pain, phantom limb pain, and limb function: a prospective study of 33 major limb amputees. Clin Orthop Relat Res. 2020;478(9):2161-2167.
- Saltzman EB, Rancy SK, Sneag DB, et al. Nerve transfers for enterovirus D68-Associated acute flaccid myelitis: a case series. Pediatr Neurol. 2018;88:25-30.
- Doi K, Sem SH, Hattori Y, et al. Contralateral obturator nerve to femoral nerve transfer for restoration of knee extension after acute flaccid myelitis. JBJS Case Connect. 2019;9(4):e0073-e0073.
- Cao Y, Li Y, Zhang Y, et al. Different surgical reconstructions for femoral nerve injury: a clinical study on 9 cases. Ann Plast Surg. 2020;84(5S Suppl 3):S171-S177.
