Current Concept Review
Management of Atypical Slipped Capital Femoral Epiphysis
1Nemours Children’s Health, Department of Orthopaedic Surgery, Wilmington, DE; 2Mount Sinai Hospital, Department of Orthopaedic Surgery, New York, NY
Correspondence: Mihir M. Thacker, MD, 1600 Rockland Road, P.O. Box 269, Wilmington, DE 19803. E-mail: [email protected]
Received: May 3, 2023; Accepted: June 22, 2023; Published: August 1, 2023
Volume 5, Number 3, August 2023
Abstract
Atypical slipped capital femoral epiphyseal (SCFE) is associated with endocrine or metabolic disorders and radiation therapy. In this review, we use case examples of hyperparathyroidism, hypothyroidism, and growth hormone deficiency, as well as renal osteodystrophy, radiation-induced, and valgus SCFE to inform the nuances of these unusual cases. From this, we learn that routine laboratory screening of “typical” patients with SCFE is not cost-effective. Patients with atypical SCFE are often short in stature, underweight, and present either older or younger than the typical age range of idiopathic SCFE. Patients fitting these criteria should undergo an endocrine workup. While uncommon, prompt recognition of atypical SCFE is crucial, as coordinated care with pediatric subspecialists is necessary. In situ fixation with cannulated screws is the most common fixation method, and bilateral fixation is recommended.
Key Concepts
- Patients younger than 10 or older than 16 years of age and patients with height, weight, or BMI below the 50th percentile should undergo laboratory screening for atypical SCFE.
- Patients presenting with a valgus slip should undergo screening labs for atypical SCFE.
- Patients presenting with a unilateral SCFE with underlying endocrinopathy, metabolic disorder, or when associated with radiation therapy should undergo bilateral fixation.
Introduction
Slipped capital femoral epiphysis (SCFE) can be categorized as either typical or atypical. Atypical SCFEs are associated with endocrine and metabolic disorders or radiation therapy.1–3 The true incidence of atypical SCFE has been reported in the range of 3%-41%, with most studies reporting the rate to be under 10%.2,4–6 In a review of available literature in 2001, Loder et al. found the average age at idiopathic SCFE diagnosis 12.8 years old. It was 15.3 years old for endocrinopathies, 11.4 years old for renal failure, and 10.5 years old for radiation therapy-associated SCFE. The distribution was wider in the atypical subgroups. Atypical SCFEs tend to present with a delayed bone age. In one descriptive study, the average bone age at presentation for atypical SCFEs was 11.6 years (average chronologic age 13.1 years) compared to a bone age of 13.1 years (average chronologic age 12 years) for typical SCFEs.6
Atypical SCFEs also have a higher proportion of bilateral involvement. The renal failure subgroup had the highest frequency of bilaterality (90%), followed by the endocrinopathies (61%), idiopathic (35%), and it was lowest in the radiation therapy-associated subgroup (28%). A simultaneous bilateral presentation was most common in the renal failure subgroup (96%) while sequential development was most frequent in the idiopathic group (49%).7 Renal failure-associated SCFEs were also more likely to present with a severe slip (40%) compared to idiopathic slips (55%), while radiation-associated SCFEs were more likely to present with a mild slip (81%) compared to idiopathic slips (55%).7 There is no significant difference in severity between endocrine-associated SCFEs and idiopathic SCFEs, or stability or chronicity between typical and atypical SCFEs.6,7
Multiple factors, both biomechanical and biochemical, are thought to contribute to the failure of the physis through the hypertrophic zone in SCFE. As children approach puberty, the physis becomes more oblique in orientation and less able to withstand shear forces with increased stress. Idiopathic SCFE can be thought of as slip due to abnormally increased force, for example, from obesity through a physiologic normal physis. Valgus SCFEs are another rare SCFE variant. The prevalence has been reported around 4% (ranging between 1.9%-9.6%).5 Valgus SCFEs present on average 2 years earlier than typical SCFEs. Multiple studies have reported a female predominance for this condition, ranging from 50%-58.3%, which has been attributed to greater femoral anteversion in females.4,5,8,9 An atypical SCFE can be conceptualized as a structurally weakened physis that can fail at physiologic loads.10 SCFE may be the first sign or symptom of an underlying pathology. In this review, we will discuss the presentation of different patients of atypical SCFEs and develop an algorithmic workup and management for an atypical SCFE.
Primary Endocrine Disorders Leading to SCFE
Hypothyroidism
Hypothyroidism is the most common endocrinopathy associated with SCFE.1,11 Patients often present with SCFE prior to hypothyroid diagnosis or treatment.1 T3 and T4 hormone receptors are found in physeal chondrocytes and play a role in their maturation.11–13 There is a reduction in the expression of aggrecan and type X collagen, leading to disorganization of the physeal architecture and loss of columnar orientation of the chondrocytes, particularly in the zone of hypertrophy.4,10 Clinical symptoms of hypothyroidism include bradycardia, fatigue, lethargy, cold intolerance, constipation, weight gain or inability to lose weight, dry skin, hair loss, memory impairment, and short stature.11,14,15 This highlights the importance of ruling out an underlying endocrinopathy, even in the obese patient. Chronic hypothyroidism can also inhibit skeletal maturation and patients can present with a delayed bone age. Many patients with hypothyroid-associated SCFE present prior to age 10, but there are a number of reported cases in older patients as well.11,14–17 Like other endocrinopathy-associated SCFEs, these patients often present with bilateral slips.10 When a child with Down Syndrome presents with a SCFE, it is important to check thyroid levels due to the association of these disorders. Pinning of both hips in these cases is advised due to the high chance of contralateral slip and the patient’s questionable ability to articulate symptoms of a subsequent slip.
Uncontrolled hypothyroidism can depress cardiac function, reduce normal responses to hypercarbia and hypoxemia, and in extreme cases, decrease lung perfusion capacity.11 There are even case reports of cardiac arrest upon induction of anesthesia in severe uncontrolled hypothyroidism.18 It is thus critical to evaluate preoperatively in the appropriate clinical scenario (as outlined previously) and if present, severe hypothyroidism should be corrected prior to surgery.
Growth Hormone Deficiency/Supplementation
Patients with growth hormone deficiency and SCFE generally present under 10 years old during or after hormone supplementation.1 The incidence of SCFE in patients taking recombinant growth hormone is more than double that of the general population.19 Growth hormone may make the physis more susceptible to shear forces.20 Animal models have indicated that growth hormone stimulates chondrocytes and proliferation in the hypertrophic zone potentially predisposing these patients to SCFE.21 The clinical manifestations of growth hormone deficiency include slow growth, short stature (<5th percentile), delayed puberty/development of secondary sexual characteristics, and headaches. Patients with conditions that lead to a deficiency in growth hormone, such as Klinefelter syndrome, may also be predisposed to SCFE.22 It is important to note, taking recombinant growth hormone (rGH) itself does not increase the risk of SCFE.23 Patients on rGH for growth hormone deficiency had a 10 times higher incidence of SCFE than those on rGH for idiopathic short stature. The average time to develop SCFE after initiating rGH therapy is 10 months.24 It is important to have a high index of suspicion for SCFE in patients taking recombinant growth hormone presenting with hip pain.
Case One
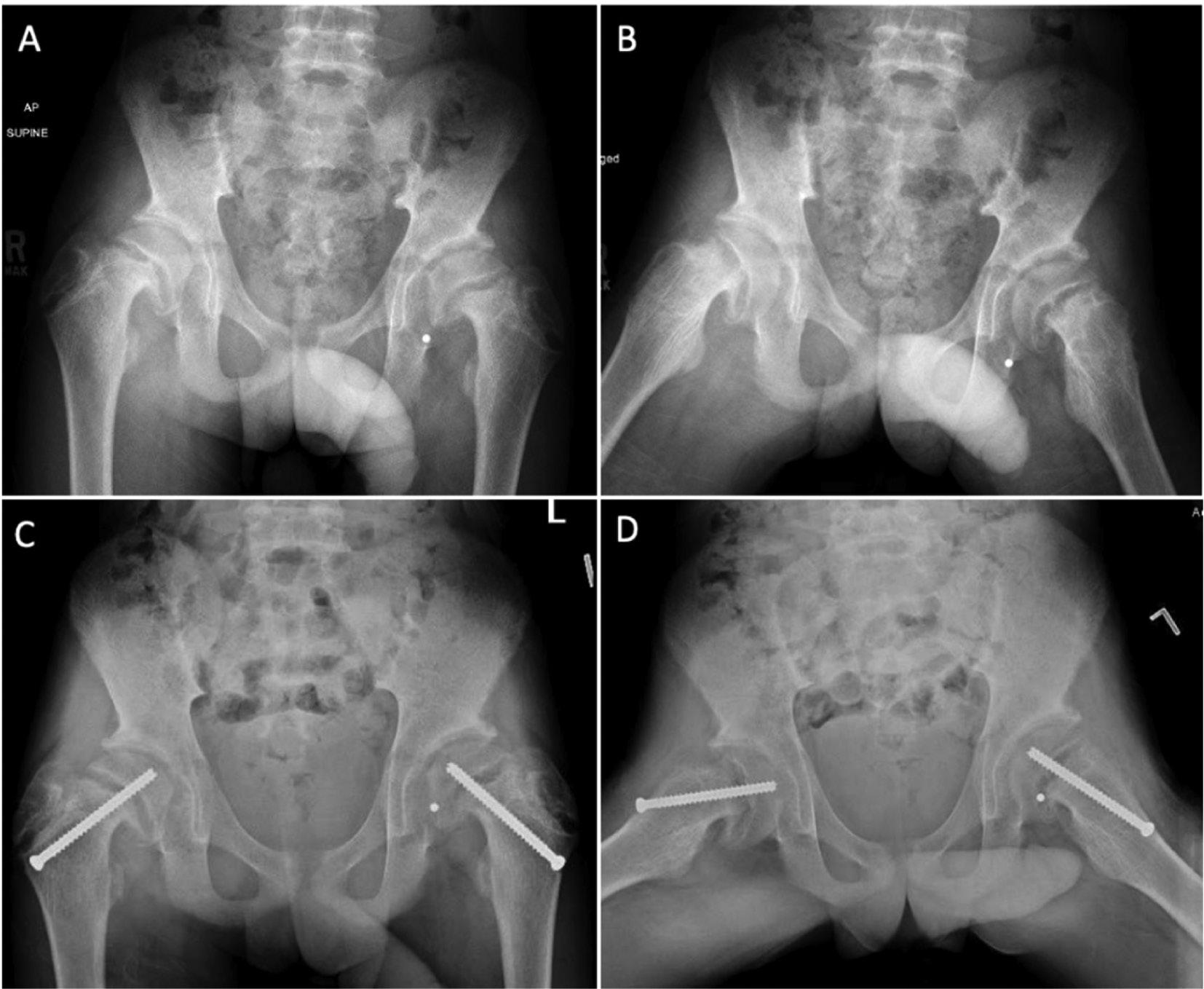
A 13-year-old male presented with a year of worsening hip pain that was limiting his ability to ambulate. He was in the 79th percentile for height, the 27th percentile for weight, and the 4th percentile for BMI. Radiographs demonstrated moderate chronic bilateral SCFE (Figure 1A, B). Laboratory screening was performed which revealed an elevated parathyroid hormone (PTH) level of 2403 PG/ML (normal: 10-65 PG/ML), mild hypocalcemia of 8.3 MG/DL (normal: 8.6-11 MG/DL), hyperphosphatemia of 6.4 MG/DL (normal: 3.6-5.6 MG/DL), and severe vitamin D-25 deficiency of < 6 NG/ML (normal: 20-50 NG/ML). He was diagnosed with hyperparathyroidism secondary to low vitamin D. He underwent bilateral in situ percutaneous fixation (Figure 1C, D), and he was started on supplemental vitamin D.
Figure 1. AP and frog leg lateral radiographs demonstrating bilateral SCFEs with physeal widening in the proximal femur and the greater trochanter as well as femoral neck sclerosis in a 13-year-old male with hyperparathyroidism pre (A-B) and post (C-D) in situ percutaneous fixation. (Images courtesy of Mihir M. Thacker, MD, Nemours Children’s Hospital, Delaware).
Hyperparathyroidism
PTH blocks normal endochondral ossification and disrupts the interdigitation that occurs between the metaphyseal bone and the physis through osteoclastic absorption, decreasing restraint to displacement, and predisposing a patient to SCFE.10,12,25,26 Clinical manifestations of hyperparathyroidism include decreased bone density, fatigue, anxiety, depression, cognitive dysfunction, constipation, hypercalciuria, nephrolithiasis, and decreased creatinine clearance (painful bones, stones, abdominal groans, and psychic moans). The most common laboratory findings are elevated PTH and hypercalcemia.27 Low levels of vitamin D may also be seen.28 These changes can manifest on radiographs or MRI as an abnormal widening or irregularity of the physis.14 Late manifestations can also include osteitis fibrosa cystica/brown tumors.29 The most common etiology of primary hyperparathyroidism is parathyroid adenoma.28
It is particularly important for urgent endocrinology consultation in these patients as profound hypercalcemia can be life-threatening, with cardiac and neurologic manifestations. Correction of severe lab abnormalities should take precedence over surgery. In the case of a parathyroid adenoma, general surgery consultation is likely required, as these are often surgically removed. The timing of orthopaedic surgical intervention depends on the severity of laboratory abnormality and should involve multidisciplinary clearance.20 Less than 20 cases of primary hyperparathyroidism have been reported in the literature, with a majority of them presenting with bilateral SCFE.28,30,31 For the cases presenting as unilateral SCFE, bilateral fixation is advocated given the risk of contralateral slip progression.26,32
A somewhat paradoxically named condition, pseudohypoparathyroidism, occurs when despite increased levels of PTH, target organ resistance to PTH results in hypocalcemia, hyperphosphatemia, thus, mimicking the clinical presentation of hypoparathyroidism.25 Pseudohypoparathyroidism-related SCFE is exceedingly rare, with only a few cases reported in the literature and all presenting with bilateral SCFE.14,33,34 Radiographic features of pseudohypoparathyroidism have been described as similar to those of hyperparathyroidism detailed above.14
Other Primary Endocrinopathies
Most endocrinopathy-related SCFEs, aside from growth hormone deficiency and some hypothyroidism, generally present when the patient is over 16 years old.1 Other endocrinopathies associated with SCFE include panhypopituitarism and hypogonadism.1,2 There are only a few case reports of panhypopituitarism-associated SCFE in the literature, with some patients presenting as late teens (16-17 years old)35,36 and others presenting as adults in the third or fourth decade of life37,38 (Figure 2). Most of these are usually followed by an endocrinologist, who can guide appropriate medical management.
Figure 2. Examples of endocrinopathy-related SCFE (A, B). Hypothyroidism with hair thinning (A, B). Panhypopituitarism (C, D). Hypogonadism in a male (testosterone deficiency) and gynecomastia (E, F).

Secondary Endocrine Disorders Leading to SCFE
Case Two
A 15-year and 8-month-old female presented with 1 year of bilateral hip pain, right worse than left. Based on her initial exam (pain with hip range of motion, lack of internal rotation, and ambulating without a limp) and radiographs (Figures 3A, B), she was presumed to have femoroacetabular impingement (FAI). However, 2 weeks later she had acute worsening of her right hip pain and inability to weight bear. Radiographs at this time demonstrated an acute-on-chronic right slip and a chronic left slip. (Figure 3). At this time, she was in the 99th percentile for height, 87th percentile for weight, and 60th percentile for BMI. Given her relatively older age and bilateral presentation, laboratory screening was pursued including a comprehensive metabolic panel, TSH, and vitamin D, which were all within normal limits.
Figure 3. Radiographs of 15-year-old female with bilateral, right acute-on-chronic, and left chronic SCFEs, who was later diagnosed with MEN2A. Radiographs from initial presentation (A-B), after acute exacerbation of right hip pain and inability to weight bear (C), following in situ percutaneous fixation (D-E), and after bilateral surgical hip dislocations with osteochondroplasty and relative femoral neck lengthening for residual femoroacetabular impingement (F-G). (Images courtesy of Mihir M. Thacker, MD, Nemours Children’s Hospital, Delaware).

She initially underwent a right hip gentle partial reduction with screw fixation and capsulotomy of the right-sided acute-on-chronic unstable SCFE as well as in situ screw fixation of the left chronic stable SCFE. Subsequently, she underwent staged bilateral surgical hip dislocations with osteochondroplasties and greater trochanteric advancements for residual FAI (Figure 3F and G). She was also found to have wrist weakness and bilateral cavovarus feet and referred to genetics for Charcot-Marie-Tooth evaluation. A targeted gene panel was negative for hereditary neuropathy. A spine MRI was obtained to evaluate for intraspinal anomalies given her cavovarus feet and development of lower extremity hyperreflexia. A thyroid nodule was seen on MRI and subsequently biopsied, which led to the diagnosis of medullary thyroid carcinoma. Labs at this time were pertinent for significantly elevated calcitonin 1,368 PG/ML (normal 0-5 PG/ML) and carcinoembryonic antigen (CEA) 12.7 NGH/ML (normal: 0-4.7 NG/ML). She was ultimately diagnosed with Multiple Endocrine Neoplasia 2A (MEN2A) syndrome due to a RET C634Y mutation and underwent a total thyroidectomy. She was treated for postoperative hypothyroidism and screened yearly for pheochromocytoma and hyperparathyroidism.
Multiple Endocrine Neoplasia
Medullary thyroid carcinoma is an uncommon pediatric diagnosis and is most commonly associated with MEN type 2A or type 2B, which results from mutations in the RET proto-oncogene. There are isolated case reports of patients presenting with MEN2B and one of MEN2A-related SCFE. Clinical findings of MEN include marfanoid body habitus, kyphoscoliosis, and pes cavus. There is a 50% risk of pheochromocytoma in both MEN 2A and 2B. MEN 2A is also associated with hyperparathyroidism while patients with MEN2B may develop mucosal neuromas on the tongue, lips, or eyelids.39–42
Other conditions resulting in secondary endocrinopathies include neoplasia (neuroblastoma, retinoblastoma, medulloblastoma, ependymoma, craniopharyngioma, and Wilms tumor). Craniopharyngioma is associated with panhypopituitarism while Turner syndrome is associated with hypogonadism (primary ovarian deficiency). Kallman syndrome is the most common genetic cause of hypogonadotropic hypogonadism.43 Optic nerve gliomas have also been described in SCFE patients.1,2,4
Metabolic Disorders Leading to SCFE
Case Three
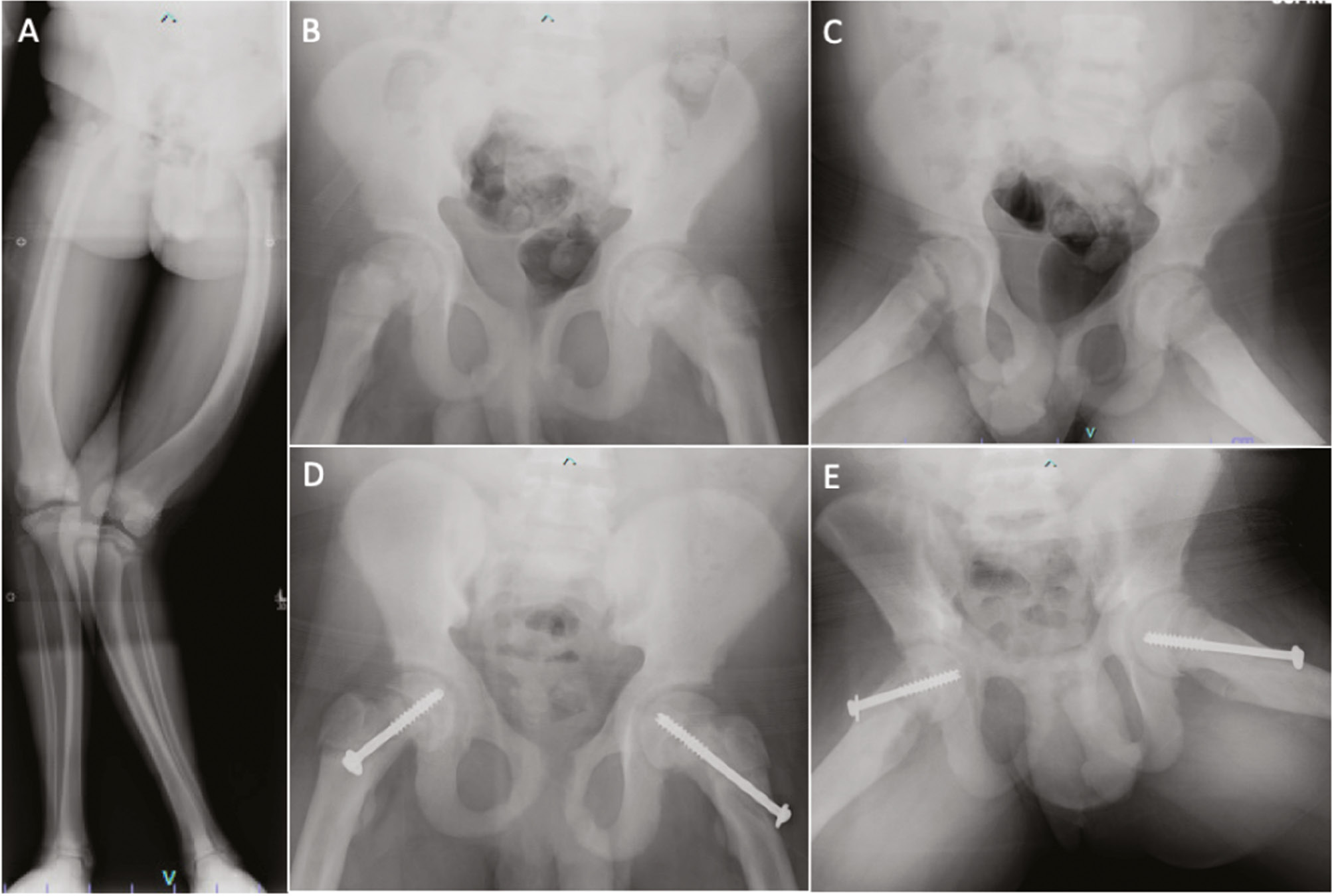
Case three is a 15-year-old African American male with renal osteodystrophy resulting from bilateral dysplastic kidneys, who underwent a failed kidney transplant and subsequently required hemodialysis. He was followed closely by the orthopaedic team for femoral bowing and genu valgum. Deformity correction was discussed, but his hyperparathyroid status was uncontrolled. He developed acute onset of right hip pain and pain with weight-bearing. Radiographs were obtained and demonstrated an acute-on-chronic moderate right SCFE and mild left SCFE. Labs were pertinent for elevated BUN 94 MG/DL (normal 5), Creatinine 12.6 MG/DL (normal <1.2), PTH 1276 PG/ML (normal 10-65), alkaline phosphatase 707 U/L (normal 92), phosphate 5.7 MG/DL (normal 2.4), potassium 6.4 MMOL/L (normal 3.6), uric acid 8.7 MG/DL (normal 2.6) as well as anemia. Following coordination and clearance by nephrology and endocrinology, he underwent bilateral in situ percutaneous fixation (Figure 4). After his hyperparathyroidism was better controlled, he underwent bilateral lower extremity deformity correction as well.
Figure 4. 15-year-old male with renal osteodystrophy resulting in lower extremity deformity, bowing and genu valgum (A), and acute moderate right SCFE and mild left SCFE at time of presentation (B-C) and following in situ fixation (D-E). (Images courtesy Mihir M. Thacker, MD, Nemours Children’s Hospital, Delaware).
Renal Osteodystrophy
End-stage renal disease leads to renal osteodystrophy which encompasses a spectrum of skeletal abnormalities including SCFE as well as characteristic radiographic findings of osteomalacia and physeal widening at the hips, knees, ankles, and wrists.44,45 While etiology of renal osteodystrophy is diverse, it leads to a downstream effect of hyperparathyroidism, which then is related to development of SCFE.46 Patients with renal osteodystrophy-associated SCFE are characteristically younger, shorter, have lower weight or BMI, and more often present with bilateral slips compared to patients with typical SCFE.4,45,46 A multidisciplinary approach with co-management by a nephrologist and an endocrinologist, both before and after surgery, is crucial, as there is evidence that failure to correct metabolic abnormalities may lead to slip progression despite surgical fixation.45 Advocated nonoperative management of patients presenting with renal failure-associated SCFE is primarily historical.25
Radiation-Induced SCFE
Case Four
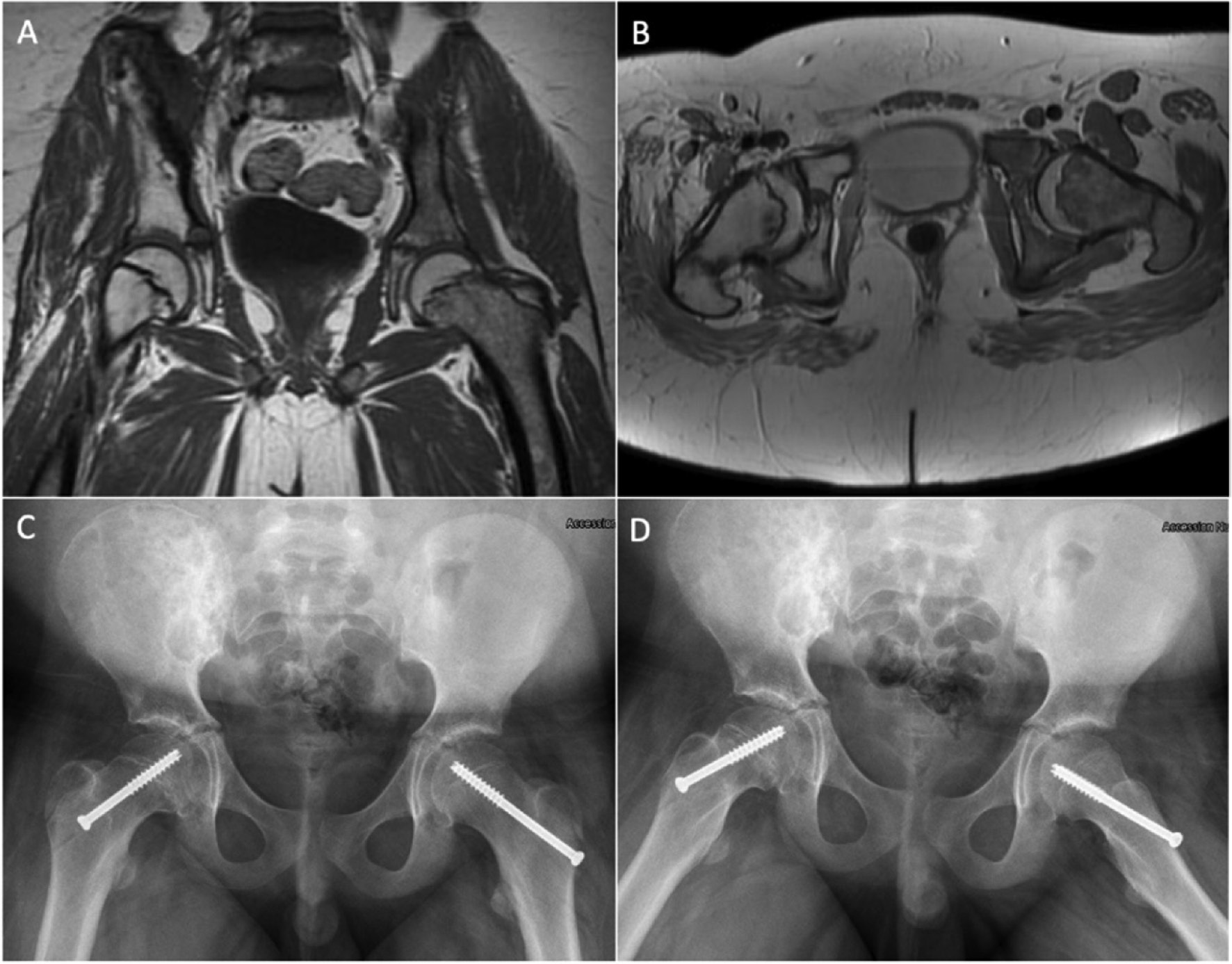
A 10-year and 1-month-old male was diagnosed with Ewing Sarcoma of the right hemipelvis after several months of right hip pain and treated with chemotherapy and local radiation. Nineteen months later, at 11 years and 8 months old, he reported right hip pain, and routine surveillance MRI demonstrated widening of the lateral posterior aspect of the right proximal physis consistent with SCFE as well as a mild left SCFE (Figure 5). He was in the 99.9th percentile for weight and BMI and 80.1st percentile for height. He was diagnosed with a mild chronic right SCFE and underwent in situ percutaneous screw fixation bilaterally (Figure 5).
Figure 5. Coronal (A) and Axial (B) MRI pelvis cuts demonstrating widening of the right posterolateral physis in a patient with post-radiation SCFE. AP (C) and frog leg lateral (D) after bilateral percutaneous fixation for post-radiation induced SCFE. (Images courtesy of Mihir M. Thacker, MD, Nemours Children’s Hospital, Delaware).
Post Radiation Therapy
Children exposed to either total body radiation or pelvic radiation have been shown to be at risk for developing subsequent SCFE. While the pathophysiology is not fully understood, it is likely a combination of direct injury to radiosensitive chondrocytes and downstream effects of sex hormone deficiency on the physis.47,48 Interestingly, an association has been previously reported between radiation and atypical valgus slips.4 In an analysis of 31 children with radiation-associated SCFE, Loder et al. found that these patients presented on average 6 years after radiation.49 The rate of patients presenting with bilateral radiation-induced SCFE is high, ranging from 28%-70% in the literature.49,50 The typical patient presenting with radiation-induced SCFE is younger and often underweight compared to idiopathic SCFE patients.49 The majority of radiation-induced SCFEs are mild at presentation.7 While the rate of contralateral slip progression is unclear in this patient population, strong consideration should be given to prophylactic fixation in patients presenting with a unilateral radiation-induced slip.
Valgus SCFE
Case Five
An 8-year and 5-month-old girl presented with 4 months of left groin and knee pain. She initially had left knee pain after a fall and only left knee radiographs were obtained by her pediatrician, which were normal. She developed left hip pain and continued to have frequent falls, limp, and difficulty ambulating, which prompted reevaluation. She was in the 99.99th+ percentile for height, weight, and BMI. Her clinical exam was positive for painful limitation of left hip range of motion, antalgic gait, and acanthosis nigricans on her neck. Radiographs demonstrated a valgus SCFE with posterior and lateral translation. Her bone age was 11 at this time. Of note, she was diagnosed with premature adrenarche at age 5 by endocrinology and prediabetes at 6 years old. Given her endocrine abnormalities, she underwent bilateral in situ screw fixation (Figure 6).
Figure 6. 8.5-year-old girl with precocious puberty, pre-diabetes, advanced bone age, and left valgus SCFE (A-B), who underwent bilateral in situ screw placement (C-D). (Images courtesy of Mihir M. Thacker, MD, Nemours Children’s Hospital, Delaware)

Valgus SCFE
Valgus slips are an uncommon variant of SCFEs where the epiphysis is translated posterolateral. Studies have shown that the femoral necks are 20 degrees more vertical than normal (153-155 degrees vs. 135 degrees), and the physes have a lateral tilt of 11 degrees compared to the normal medial tilt of 16 degrees.5,51 The degree of posterior physeal tilt reported for valgus slips is similar to that of conventional SCFEs. Biomechanical studies found the physeal shear force in valgus slips to be 2.7 times body weight in the posterolateral direction compared to 2.2 times body weight in typical SCFE.51 In a recent meta-analysis of patients with valgus slips, 22% (7/32) had an underlying endocrine disorder.5 Chung et al. reported on 22 patients with 29 atypical SCFEs with a majority secondary to radiation or chemotherapy treatment. They found that 76% (22/29) of these slips were a valgus SCFE. Their analysis is one of the largest cohorts of patients presenting with valgus slips.2 A smaller single-center analysis of valgus SCFE patients found that 42% (5/12) valgus slips were associated with an atypical SCFE.5
Data on the risk of contralateral slip progression for patients with valgus SCFE is limited. Gelink et al. reported on a small case series of 8 patients with valgus SCFE: 2/8 presented as bilateral slips, 1 patient subsequently developed a contralateral slip, and all of the patients were thought to have idiopathic SCFE.52 A larger metanalysis found that 31% of valgus SCFE patients (23/74) initially presented as a bilateral SCFE, which is higher than the rate of 18% that has been reported for idiopathic SCFE.9,53
While valgus slips represent an uncommon subset of SCFE, with about 100 reported cases in the literature, their unique radiographic characteristics make it important for us to identify them. Patients presenting with a valgus SCFE should undergo screening lab work given the high proportion of valgus slip associated with endocrine or metabolic abnormalities.5,9
Screening Recommendations for Orthopaedic Providers
Loder et al. described and provided validation for the age-weight and age-height tests—tools that can guide orthopaedists in identifying patients with atypical SCFE.6,7 Loder found that children younger than 10 or older than 16 were 7.4 times more likely to have an atypical SCFE. Furthermore, children presenting with weight less than the 50th percentile were six times more likely to have an atypical SCFE while children with height less than the 50th percentile were 13.1 times more likely.6 A negative age-height (10-16 years old, 50th%+ height) and negative age-weight (10-16 years old, 50th%+ height) had a 98% and 94% negative predictive value for atypical SCFE, respectively. Similarly, the height test proposed by Burrow, based on height less than the 10th percentile, had a negative predictive value of 97%.54 The sensitivity of the Loder age-height test and the Burrow height tests were 88% and 75%, respectively. Height appears to be the most useful characteristic when ruling out atypical SCFE. An attempt should be made to obtain a current height and weight for every patient presenting with a SCFE, even by history of the last PCP visit. Given that most patients with endocrine or metabolic-associated SCFE, except for hypothyroidism, often present as underweight, orthopaedists should be suspicious of underweight patients with a SCFE.
We recommend patients younger than 10 or older than 16, presenting with a height below the 50th percentile or weight below the 50th percentile, bilateral SCFE, or a valgus SCFE undergo screening for underlying disorders6,54 (Figure 7). Screening lab work for underlying endocrine and metabolic conditions should start with TSH, PTH, and a comprehensive metabolic panel. If TSH is elevated, a full thyroid workup, including free thyroxine, should be performed. If PTH is abnormal, vitamin D levels should be obtained (Table 1). Endocrine referral should be strongly considered in patients with atypical presentation (age-weight test, valgus slip, suspicious history/clinical evaluation, delayed bone age, atypical radiographic exam) but have a negative initial screen to further evaluate growth hormone and gonadal hormone-related disorders. We also utilize our endocrinology colleagues to help in multidisciplinary management of patients with known endocrinopathies and those with a positive initial screen. Patients presenting with known renal osteodystrophy-associated SCFE should undergo laboratory screening for hyperparathyroidism. To our knowledge, no studies to date have directly examined the cost-effectiveness of screening labs in patients presenting with SCFE, but as outlined above, it is crucial to identify these conditions to avoid associated morbidity or mortality.
Figure 7. Treatment algorithm for the workup of atypical SCFE at presentation.
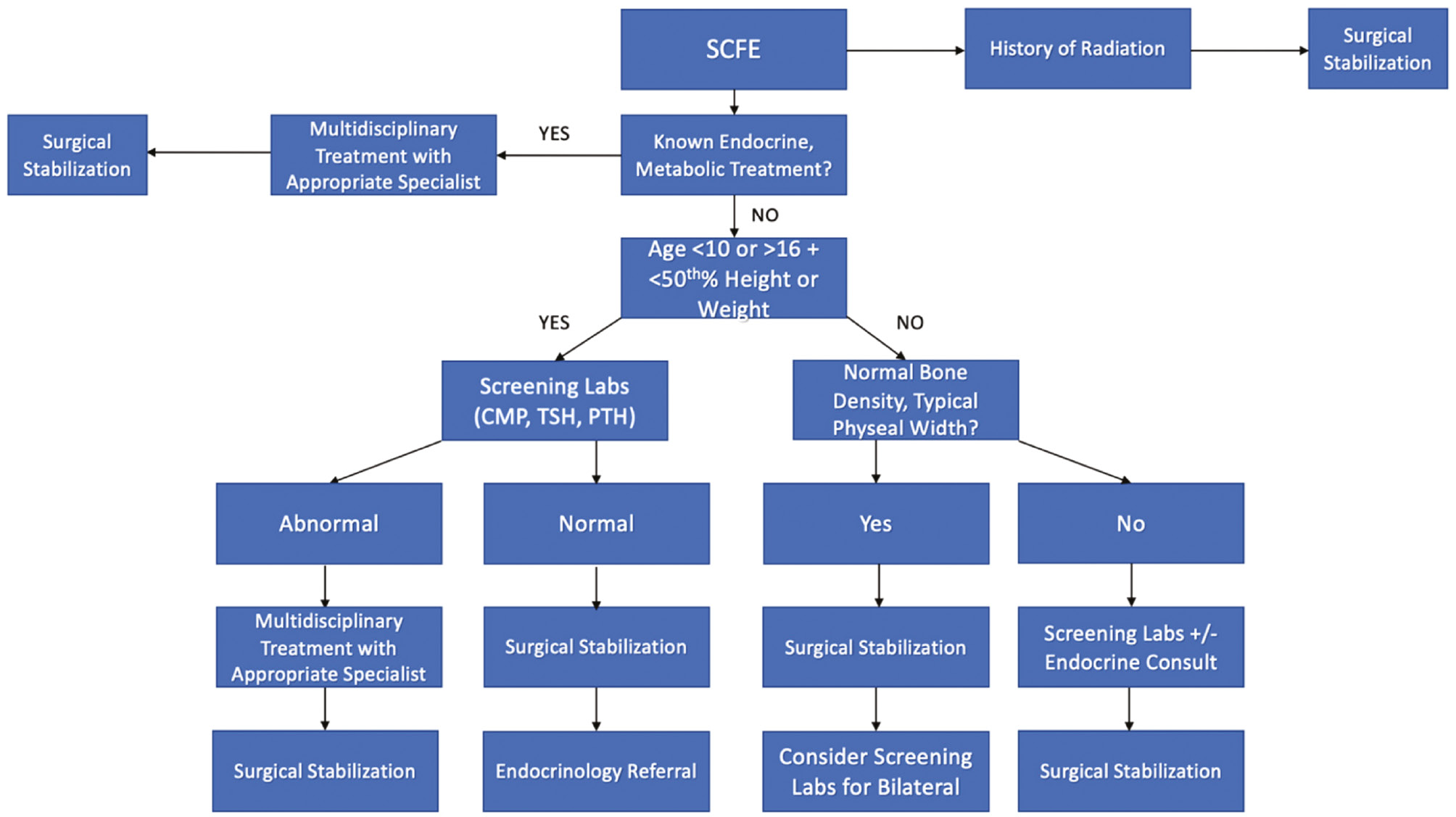
Table 1. Lab Abnormalities and Treatment for Endocrinopathies
| Disorder | Laboratory Derangements | Treatment |
|---|---|---|
| Hypothyroidism | ↑TSH, ↓T4 | Thyroxine replacement |
| Growth Hormone Deficiency | ↓GH | Recombinant GH |
| Hyperparathyroidism | ↑PTH, ↑Ca, +/- ↓Vit D | Parathyroidectomy, Vit D supplementation |
| Hypogonadism | Primary: ↓Estrogen/Testosterone, ↑FSH/LH Secondary: ↓Estrogen/ Testosterone, ↓FSH/LH |
Hormone replacement therapy Further Workup Secondary Hypogonadism indicates hypothalamic or pituitary dysfunction |
| Panhypopituitarism | ↓TSH, ↓GH, ↓ACTH, ↓FSH, ↓LH, ↓prolactin | Hormone replacement therapy +/- surgery, radiation therapy |
| MEN2A/2B | ↑calcitonin, ↑CEA, +/-↑PTH | Thyroidectomy (medullary thyroid carcinoma) Screen for hyperparathyroidism/pheochromocytoma |
| Renal Osteodystrophy | ↑BUN, ↑Cr, ↑phos, ↑PTH, ↓Ca | Dialysis, Ca supplementation, Calcimimetics Screen for hyperprarathyoidism |
All patients with lab abnormalities or an identified endocrinopathy should be referred to specialists for all treatment and management of underlying pathology.
Consideration should also be given to testing and supplementing vitamin D if deficient. Although there is conflicting evidence in the literature, with one study showing no association between vitamin D deficiency and SCFE,55 others have reported 85%-100% vitamin D deficiency in their SCFE cohorts.56,57 Judd et al., also found the time to physeal fusion was prolonged to 20 months in SCFE patients with vitamin D deficiency compared to 9.6 months in controls.56 Vitamin D supplementation should be started if a patient is found to be deficient.
Management
If an underlying endocrine or metabolic abnormality is identified, the appropriate medical specialists should be consulted for treatment and to determine if the patient is stable enough for surgery. Surgical fixation options are the same as for idiopathic SCFEs. The majority are fixed with in situ cannulated screw fixation. In patients with extremely soft bone, the use of a washer may be considered (Figure 4). There is no published literature comparing one or two screw fixation within the atypical SCFE population. We recommend the determination for one versus two screws be made based on slip stability, as two screws are biomechanically more stable and may be warranted for unstable slips.58 Gentle closed reduction (+/- capsulotomy) can be attempted in cases of an unstable SCFE. Other initial treatment options include open reduction using an anterior approach or the modified Dunn technique for surgeons facile in this technique.59–61
Patients presenting with a unilateral slip who are diagnosed with an underlying endocrinopathy or renal disease should undergo bilateral in situ screw fixation given the high risk of contralateral slip progression. Patients who develop radiotherapy-associated unilateral atypical SCFE, who are found to have a co-existing endocrinopathy, should undergo contralateral fixation as well. The risk of contralateral slip progression in a patient with isolated radiation-associated SCFE is less clear. Pelvic or full-body radiation can weaken bilateral proximal femoral physes, and bilateral fixation should be strongly considered. All patients presenting with bilateral SCFE, whether typical or atypical, should undergo bilateral fixation (Figure 6).
Special consideration should be given to the fixation of valgus SCFE. The trajectory for a percutaneous guidewire may be very medial/anterior and potentially put the femoral neurovascular bundle at risk. Some have recommended a limited open anteromedial approach with identification and protection of the neurovascular bundle5 (Figure 8).
Figure 8. Valgus SCFE screw trajectory noted by white line. The more valgus the slip, the closer the entry point is to the neurovascular bundle. (Images courtesy of Mihir M. Thacker, MD, Nemours Children’s Hospital, Delaware).

No studies have specifically reported a higher rate of complications or slip progression after in situ fixation in atypical slips compared to typical slips. Makarewich et al. found that less than three threads across the physis were more predictive of failure and there with no increased rate of failure in endocrine SCFEs within their cohort compared to typical SCFEs.62 Similarly younger age has been associated with increased risk of slip. In one study, the average age of those with slip progression was 10.6 years compared to an average age of 12.7 years in the successful treatment group.63 It is also important to make sure the underlying pathology remains controlled. In a study of outcomes for renal osteodystrophy-associated SCFE, 88% of slips stabilized after surgery, and those that progressed had poorly controlled renal disease necessitating further medical management.45
Summary
Atypical SCFE is associated with underlying endocrinopathies, metabolic disorders such as renal osteodystrophy, and radiation therapy. Given the relatively low frequency and sometimes subtle presentation, the diagnosis of atypical SCFE can be challenging. A systemic approach to all patients presenting with a SCFE is crucial and should begin with a thorough history and physical exam. An accurate height and weight should be obtained if possible. We recommend all patients with either a positive age-height or age-weight test, bilateral or valgus SCFE presentation undergo a screening endocrine workup consisting of a comprehensive metabolic panel, PTH and TSH, which can be further tailored pending pertinent derangements Vitamin D evaluation should considered, especially in areas with endemic Vitamin D deficiency. Patients with undiagnosed endocrinopathies or renal disease need prompt referral and medical management from pediatric specialists. Surgical fixation options remain the same as for idiopathic SCFE; however, atypical SCFE cases benefit from prophylactic fixation of the contralateral side as well.
Disclaimer
The authors report no conflicts of interest related to this manuscript.
References
- Loder RT, Wittenberg B, Desilva G. Slipped capital femoral epiphysis associated with endocrine disorders. J Pediatr Orthop. 1995;15(3):349-356.
- Wells D, King JD, Roe TF, et al. Review of slipped capital femoral epiphysis associated with endocrine disease. J Pediat Orthop. 1993;13(5):610-614.
- McAfee PC, Cady RB. Endocrinologic and metabolic factors in atypical presentations of slipped capital femoral epiphysis. Report of four cases and review of the literature. Clin Orthop Relat Res. 1983;(180):188-197.
- Chung CH, Ko KR, Kim JH, et al. Clinical and radiographic characteristics of atypical slipped capital femoral epiphysis. J Pediatr Orthop. 2019;39(10):E742-E749.
- Shank CF, Thiel EJ, Klingele KE. Valgus slipped capital femoral epiphysis: prevalence, presentation, and treatment options. J Pediatr Orthop. 2010;30(2):140-146.
- Loder RT, Starnes T, Dikos G. Atypical and typical (Idiopathic) slipped capital femoral epiphysis. J Bone Joint Surg. 2006;88(7):1574-1581.
- Loder RT, Greenfield MLVH. Clinical characteristics of children with atypical and idiopathic slipped capital femoral epiphysis: description of the age-weight test and implications for further diagnostic investigation. J Pediatr Orthop. 2001;21(4):481-487.
- Koczewski P. Valgus slipped capital femoral epiphysis: subcapital growth plate orientation analysis. J Pediatr Orthop B. 2013;22(6):548-552.
- Assi C, Mansour J, Kouyoumdjian P, et al. Valgus slipped capital femoral epiphysis: a systematic review. J Pediatr Orthop B. 2021;30(2):116-122.
- Witbreuk M, van Kemenade FJ, van der Sluijs JA, et al. Slipped capital femoral epiphysis and its association with endocrine, metabolic and chronic diseases: a systematic review of the literature. J Child Orthop. 2013;7(3):213-223.
- Moyer J, Jacks L, Hunter JD, et al. Slipped capital femoral epiphysis and associated hypothyroidism. A review of the literature with two classic case examples. J Pediatr Endocrinol Metab. 2016;29(4):427-434.
- Krempien B, Mehls O, Ritz E. Morphological studies on pathogenesis of epiphyseal slipping in uremic children. Virchows Arch A Pathol Anat Histol. 1974;362(2):129-143.
- Gogakos AI, Duncan Bassett JH, Williams GR. Thyroid and bone. Arch Biochem Biophys. 2010;503(1):129-136.
- Zelaya R, Zarka A, Byerly D. Slipped capital femoral epiphysis as a presentation of underlying metabolic disorders: pseudohypoparathyroidism and juvenile hypothyroidism. Cureus. 2021;13(3):1-7.
- Kadowaki S, Hori T, Matsumoto H, et al. Prepubertal onset of slipped capital femoral epiphysis associated with hypothyroidism: a case report and literature review. BMC Endocr Disord. 2017;17(1):1-5.
- Hennessy MJ, Jones KL. Slipped capital femoral epiphysis in a hypothyroid adult male. Clin Orthop Relat Res. 1982;165:204-207.
- Marquez D, Harb E, Vilchis H. Slipped capital femoral epiphysis and hypothyroidism in a young adult: a case report. J Med Case Rep. 2014;8(1):3-6.
- Awari DW, Dhanasekaran M, Mudrakola HV, et al. Myocardial infarction and cardiac arrest in a patient with severe undiagnosed hypothyroidism during bronchoscopy. J Cardiothorac Vasc Anesth. 2022;36(5):1410-1414.
- Blethen SL, Allen DB, Graves D, et al. Safety of recombinant deoxyribonucleic acid-derived growth hormone: The National Cooperative Growth Study experience. J Clin Endocrinol Metab. 1996;81(5):1704-1710.
- Harris WR, Mehls O, Ritz E, et al. Primary hyperparathyroidism presenting as slipped capital femoral epiphysis. J Child Orthop. 2022;96(1):1-5.
- Docquier PL, Mousny M, Jouret M, et al. Orthopaedic concerns in children with growth hormone therapy. Acta Orthop Belg. 2004;70(4):299-305.
- Song KS, Lim YW, Ok IY, et al. Delayed-onset of slipped capital femoral epiphysis. J Orthop Sci. 2015;20(1):78-86.
- Allen DB. Safety of growth hormone treatment of children with idiopathic short stature: the US experience. Horm Res Paediatr. 2011;76(Suppl 3):45-47.
- Blethen SL, Rundle AC. Slipped capital femoral epiphysis in children treated with growth hormone. A summary of the National Cooperative Growth Study experience. Horm Res. 1996;46(3):113-116.
- Mehls O, Ritz E, Krempien B, et al. Slipped epiphyses in renal osteodystrophy. Arch Dis Child. 1975;50(7):545-554.
- Bone LB, Roach JW, Ward WT, et al. Slipped capital femoral epiphysis associated with hyperparathyroidism. J Pediatr Orthop. 1985;5(5):589-592.
- Zhu CY, Sturgeon C, Yeh MW. Diagnosis and management of primary hyperparathyroidism. JAMA. 2020;323(12):1186-1187.
- Vitale RJ, Shieh HF, Modi BP, et al. Primary hyperparathyroidism from ectopic parathyroid adenoma in a 12-year-old with slipped capital femoral epiphysis. J Endocr Soc. 2022;6(6):1-5.
- El Scheich T, Marquard J, Westhoff B, et al. Approach to the management of slipped capital femoral epiphysis and primary hyperparathyroidism. J Pediatr Endocrinol Metab. 2012;25(5-6):407-412.
- George G, Raizada N, Jabbar P, et al. Slipped capital femoral epiphysis in primary hyperparathyroidism - Case report with literature review. Indian J Endocrinol Metab. 2019;23(4):491-494.
- Roztoczyńska D, Wójcik M, Konturek A, et al. Bilateral slipped capital femoral epiphysis as first manifestation of primary hyperparathyroidism in a 15-year-old boy. Pediatr Endocrinol Diabetes Metab. 2020;26(4):220-224.
- Kumar G, Mathew V, Kandathil JC, et al. Primary hyperparathyroidism presenting as slipped capital femoral epiphysis. Postgrad Med J. 2020;96(1134):235-236.
- Somasundaram KR, Sankararaman S, Siddiqui A, et al. Pseudohypoparathyroidism as a rare cause of bilateral slipped capital femoral epiphysis. Indian J Orthop. 2012;46(6):705-709.
- Agarwal C, Seigle R, Agarwal S, et al. Pseudohypoparathyroidism: a rare cause of bilateral slipped capital femoral epiphysis. J Pediatr. 2006;149(3):406-408.
- Harris JB, Sunil B, Ryan MK, et al. Association of slipped capital femoral epiphysis with panhypopituitarism due to pituitary macroadenoma: a case report. J Investig Med High Impact Case Rep. 2021;9:2324709621999956.
- Bowden SA, Klingele KE. Chronic bilateral slipped capital femoral epiphysis as an unusual presentation of congenital panhypopituitarism due to pituitary hypoplasia in a 17-year-old female. Int J Pediatr Endocrinol. 2009;2009:609131.
- Rosen M, Wong A, Worts P, et al. Slipped capital femoral epiphysis in an adult with panhypopituitarism: a case report. JBJS Case Connect. 2021;11(3). http://doi.org/10.2106/JBJS.CC.20.00940.
- Niu Z, Tang J, Shen X, et al. Slipped capital femoral epiphysis with hypopituitarism in adults: a case report and literature review. Medicine. 2021;100(51):e28256.
- Ahmad M, Rizvi I, Jain A, et al. Painful hip leading to the diagnosis of MEN 2B syndrome. Case Rep Endocrinol. 2012;2012:1-3.
- Beukhof CM, van Biezen FC, de Herder WW. Slipped capital femoral epiphysis as manifestation of a rare endocrinological disease. Neth J Med. 2011;69(2):84-94.
- Saltzman CL, Herzenberg JE, Phillips WA, et al. Thick lips, bumpy tongue, and slipped capital femoral epiphysis--a deadly combination. J Pediatr Orthop. 1988;8(2):219-222.
- Gladstein AZ, van Dijck S, Wasserman JD, et al. Painful hips and a nodular neck: bilateral slipped capital femoral epiphysis leading to the diagnosis of multiple endocrine neoplasia: a case report. JBJS Case Connect. 2015;5(4):e106.
- Richard-Eaglin A. Male and female hypogonadism. Nurs Clin North Am. 2018;53(3):395-405.
- Moe S, Drüeke T, Cunningham J, et al. Definition, evaluation, and classification of renal osteodystrophy: a position statement from Kidney Disease: improving Global Outcomes (KDIGO). Kidney Int. 2006;69(11):1945-1953.
- Oppenheim WL, Bowen RE, McDonough PW, et al. Outcome of slipped capital femoral epiphysis in renal osteodystrophy. J Pediatr Orthop. 2003;23(2):169-174.
- Loder RT, Hensinger RN. Slipped capital femoral epiphysis associated with renal failure osteodystrophy. J Pediatr Orthop. 1997;17(2):205-211.
- Kawakami K, Saito T, Tauchi R, et al. Nonanesthetized alternatively repetitive cast and brace treatment for early-onset scoliosis. J Pediatr Orthop. 2020;40(8):e720-e727.
- Riseborough EJ, Grabias SL, Burton RI, et al. Skeletal alterations following irradiation for Wilms’ tumor: with particular reference to scoliosis and kyphosis. J Bone Joint Surg Am. 1976;58(4):526-536.
- Loder RT, Hensinger RN, Alburger PD, et al. Slipped capital femoral epiphysis associated with radiation therapy. J Pediatr Orthop. 1998;18(5):630-636.
- Mostoufi-Moab S, Isaacoff EJ, Spiegel D, et al. Childhood cancer survivors exposed to total body irradiation are at significant risk for slipped capital femoral epiphysis during recombinant growth hormone therapy. Pediatr Blood Cancer. 2013;60(11):1766-1771.
- Yngve DA, Moulton DL, Burke Evans E. Valgus slipped capital femoral epiphysis. J Pediatr Orthop B. 2005;14(3):172-176.
- Gelink A, Cúneo A, Silveri C, et al. Valgus slipped capital femoral epiphysis: presentation, treatment, and clinical outcomes using patient-reported measurements. J Pediatr Orthop B. 2021;30(2):111-115.
- Loder RT, Aronson DD, Greenfield ML. The epidemiology of bilateral slipped capital femoral epiphysis. A study of children in Michigan. J Bone Joint Surg Am. 1993;75(8):1141-1147.
- Burrow SR, Alman B, Wright JG. Short stature as a screening test for endocrinopathy in slipped capital femoral epiphysis. J Bone Joint Surg B. 2001;83(2):263-268.
- Arkader A, Woon RP, Gilsanz V. Can subclinical rickets cause SCFE? A prospective, pilot study. J Pediatr Orthop. 2015;35(7):e72-e75.
- Judd J, Welch R, Clarke A, et al. Vitamin D deficiency in slipped upper femoral epiphysis: time to physeal fusion. J Pediatr Orthop. 2016;36(3):247-252.
- Madhuri V, Arora SK, Dutt V. Slipped capital femoral epiphysis associated with vitamin D deficiency: a series of 15 cases. Bone Joint J. 2013;95-B(6):851-854.
- Schmitz MR, Farnsworth CL, Doan JD, et al. Biomechanical testing of unstable slipped capital femoral epiphysis screw fixation: worth the risk of a second screw? J Pediatr Orthop. 2015;35(5):496-500.
- Tannast M, Jost LM, Lerch TD, et al. The modified Dunn procedure for slipped capital femoral epiphysis: the Bernese experience. J Child Orthop. 2017;11(2 Special Issue):138-146.
- Haider S, Podeszwa DA, Morris WZ. The etiology and management of slipped capital femoral epiphysis. J Pediatr Orthop Soc N Am. 2022;4(4). http://doi.org/10.55275/jposna-2022-589
- Napora JK, Morris WZ, Gilmore A, et al. Purposeful closed reduction and pinning in unstable slipped capital femoral epiphysis results in a rate of avascular necrosis comparable to the literature mean. Orthopedics. 2021;44(2):92-97.
- Makarewich CA, Wait ES, Fedorak GT, et al. Factors affecting slip progression after in situ screw fixation of stable slipped capital femoral epiphysis. J Pediatr Orthop. 2022;42(5):e414-e420.
- Carney BT, Birnbaum P, Minter C. Slip progression after in situ single screw fixation for stable slipped capital femoral epiphysis. J Pediatr Orthop. 2003;23(5):584-589.
