2023 POSNA Annual Meeting – Best Basic Science Research Paper
Like a Lizard: Repair of Perthes Disease Occurs Through Recapitulation of Development of the Proximal Femoral Epiphysis
1Vanderbilt University Medical Center, Department of Orthopaedics, Monroe Carrol Jr. Children’s Hospital at Vanderbilt, Division of Pediatric Orthopaedics, Nashville, TN; 2Vanderbilt University Medical Center, Department of Orthopaedics, Nashville, TN; 3Vanderbilt University Medical Center, Department of Pathology, Microbiology, and Immunology, Monroe Carrol Jr. Children’s Hospital at Vanderbilt, Nashville, TN
Correspondence: Jonathan G. Schoenecker, MD, PhD, Vanderbilt University Medical Center, Department of Orthopaedics, Division of Pediatric Orthopaedics, 2200 Children’s Way, Nashville TN 37232. E-mail: [email protected]
Received: May 16, 2023; Accepted: May 16, 2023; Published: August 1, 2023
Volume 5, Number 3, August 2023
Abstract
Introduction: Legg-Calve-Perthes Disease (LCPD) is a complex condition with limited understanding about its healing process. The healing mechanism of LCPD is believed to differ significantly from adult hip avascular necrosis (AVN), thus necessitating an exploration into alternative mechanisms. Our research focused on understanding these mechanisms using a review of histologic specimens, a thorough literature review, and translational basic science studies.
Methods: Histologic Review: IRB-approved biopsies from five Stage 2 LCPD cases undergoing hinged abduction were analyzed to confirm the presence of cartilage in areas of fragmentation. Literature Search: A comprehensive literature review was performed, scrutinizing publications on the role of cartilage during Stage 2 of LCPD from Perthes’ original observations to subsequent studies. Mouse Model: A mouse hip model, chosen for its resemblance to Stage 2 LCPD, was used to study chondrocyte-derived VEGF’s role in angiogenesis and ossification. VEGF blockade was employed to confirm its function.
Results: Histologic Review: All five biopsies of Stage 2 LCPD cases showed robust cartilage presence in areas of fragmentation (Figure 1). Histological analysis confirmed cartilage leading to new bone formation areas, akin to the physis’ zone of ossification. Literature Search: Our literature review identified consistent documentation of cartilage presence during Stage 2 of LCPD in past studies dating back to Perthes, but the role of this cartilage in the healing process was previously not clearly defined. Mouse Model: Chondrocytes were observed to produce a significant amount of VEGF right before vascularization of the hip. Blockade of VEGF production halted both vascularization and ossification. These findings point towards a role for chondrocyte-derived VEGF in promoting angiogenesis and ossification of the femoral head.
Figure 1. Representative biopsy of a patient in stage 2 LCPD (A) reveals complete chondrification of the proximal epiphysis (B) with biopsy sections (C) demonstrating cartilage laden with chondrocytes in different stages of development. Histology (D) reveals abundant chondrocytes with areas, anatomically remote from the physis, developing areas resembling the zone of ossification of the physis.
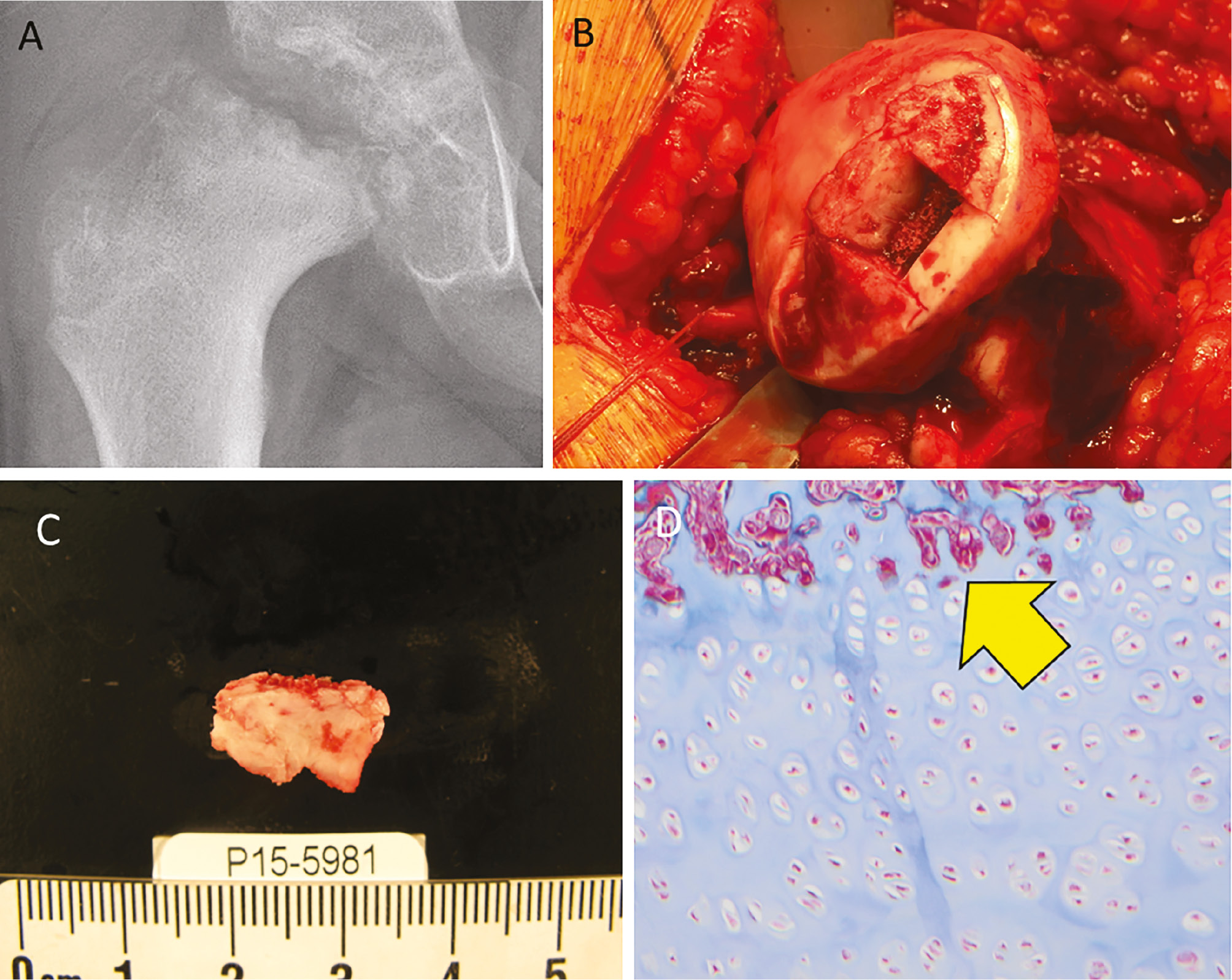
Conclusion: These findings support the theory that chondrification during Stage 2 of LCPD is part of the unique reparative process of LCPD. The production of VEGF by chondrocytes appears to be integral for vascularization and ossification of the femoral head. We propose that the healing mechanism of LCPD is not a superior form of creeping substitution, but rather an endochondral mediated mechanism of vascularization and ossification, much like a lizard regrowing its tail.
Significance: These findings clarify the healing process of LCPD and can inform future treatment protocols. Treatment protocols should consider these biological processes to optimize outcomes in LCPD. Further studies are required to fully elucidate this process and its implications on the management of LCPD. Specifically, it emphasizes the importance of supporting chondrification and the production of VEGF rather than focusing solely on anti-resorptive and osteogenics.
Acknowledgement: Special thank you to Perry Schoenecker, MD; Stuart Weinstein, MD; and Katherine Hajdu, BS.
Disclaimer: The authors report no conflicts of interest related to this abstract.
