Current Concept Review
The Evolution of Guided Growth for Lower Extremity Angular Correction
Texas Scottish Rite Hospital for Children, Dallas, TX
Correspondence: Elizabeth W. Hubbard, MD, Scottish Rite for Children, 5700 Dallas Parkway, Frisco, TX 75034. E-mail: [email protected]
Received: May 29, 2023; Accepted: June 20, 2023; Published: August 1, 2023
Volume 5, Number 3, August 2023
Abstract
Guided growth is one of the most utilized surgical techniques for managing limb deformity in skeletally immature patients. Our understanding of this technique and the types of implants utilized have evolved over the past century. Many of the known risks of this surgery, such as over-correction, under-correction, and rebound deformity, are the same risks initially described when hemiepiphysiodesis and guided growth techniques were first published. The staple has been a powerful tool for deformity correction but its high rates of implant backout and breakage as well as unpredictable rates of premature physeal closure after removal have contributed to this implant being used less frequently today. Many studies on percutaneous transepiphyseal screws (PETS) are promising but have little follow-up, so the risks of this technique with regard to premature physeal closure are not well understood. Tension band plating is currently the most utilized method. However, in specific patient subgroups, the perioperative complication and failure of correction rates are high. Despite the abundance of literature on these techniques, our understanding of guided growth is still quite limited, as most studies are small and do not follow patients to skeletal maturity. Guided growth surgery also can restore the mechanical axis of the limb while leaving patients with significant residual diaphyseal or peri-articular deformity and the implications of these secondary deformities have not been studied.
Key Concepts
- An understanding of the patient’s skeletal maturity and predicted growth remaining is essential prior to performing guided growth surgery.
- Regular patient follow-up is critical to prevent over-correction of deformity.
- Staples are an effective implant for angular deformity correction but have higher rates of implant backout, breakage, and premature physeal arrest compared to other devices.
- Percutaneous transepiphyseal screws can be an effective way to correct angular deformity, but current studies are both small and lack long-term follow-up, which makes it difficult to truly understand the potential risks and complications of this technique.
- Tension band plating is a safe and effective treatment for angular deformity correction but the rates of implant breakage and peri-surgical complications in patients with Blount disease are consistently high.
Introduction
Regardless of the etiology, significant coronal plane deformity can contribute to long-term pathology of the knee and lower extremity. Untreated varus deformity has been identified as a risk factor for degenerative arthritis.1 Genu valgum can contribute to anterior knee pain, altered patellofemoral mechanics, and is a known risk factor for patellofemoral instability.2–7 In addition, those with a unilateral compartment injury, such as meniscal derangement or osteochondral injury, can benefit from off-loading the compartment to prevent the progression of damage or to protect surgical reconstruction.1
There are multiple ways to address coronal plane deformity. The most direct is an acute osteotomy with internal fixation. While this leads to immediate correction, it may not always be the most appropriate choice for the patient. There are limits to the amount of deformity which can be safely treated in the acute setting. Acute osteotomies, particularly valgus-producing osteotomies, can also place neurovascular structures at risk at the time of correction.8
Larger deformities, particularly multiplanar deformities, are often best treated through osteoplasty and gradual correction using a multiplanar external fixator. This allows the surgeon to address deformities in the coronal, sagittal, and axial plane. Rigid fixation allows for immediate weight-bearing, and the strategic use of wires and half-pins allows for deformity correction even in patients with compromised soft tissue.9,10 However, the fixator may be in place for weeks to months while the regenerate consolidates. Pin and wire sites are each a potential nidus for infection.11 Patients also have concerns regarding the cosmesis of the pin and wire sites after removal, difficulty with getting dressed, and other activities of daily living as well as difficulty with mobility, particularly immediately after frame application.10
Guided growth, or growth modulation, can also be a powerful corrective option for skeletally immature patients. Guided growth utilizes the patient’s growth potential to correct coronal and/or sagittal plane deformity. Multiple surgical techniques have been described. Although the surgical steps differ, these surgeries are performed through relatively small incisions, patients are allowed to weight bear immediately, and overall recovery and return to activity is faster compared with osteotomy. Because the procedure relies on the patient’s skeletal growth potential to achieve deformity correction, understanding the patient’s skeletal maturity status is important. Two of the greatest risks specific to this procedure are under- or over-correction of deformity.12 Under-correction is possible if a patient is approaching skeletal maturity at the time of surgery. These patients and families need to be aware that complete deformity correction could require additional surgery. Patients who are very skeletally immature are at risk for over-correction, which could be severe enough to require additional surgery, so the need for regular outpatient follow-up visits needs to be emphasized to the family.
The surgical techniques and available implants for guided growth have evolved over the past 90 years. During this time, surgeons have started pushing the limits of when and how to perform guided growth. This procedure has been utilized for patients with underlying skeletal dysplasias, metabolic abnormalities, and deformity induced by underlying genetic and neurologic disorders.12–14 In addition, its use has been extended from controlling the coronal plane to controlling the sagittal plane and also explored as a reversible way to address leg length differences.15–24 This article will review the history of guided growth specifically for the treatment of angular deformity in the coronal plane around the knee.
Phemister Epiphysiodesis and Hemiepiphysiodesis
Prior to the 1930s, surgical options to address leg length differences were limited to either shortening the longer extremity or attempted lengthening of the shorter extremity. However, in 1927, Gatewood and Mullen reported that the longitudinal growth of a long bone was halted if the epiphyseal plate (the physis) was fused with the diaphysis of the bone at a young age.25 They reported that significant length differences were created if the entire physis was fused, but fusion of only a portion of the physis could induce an angular deformity. Phemister extended this work and described the bone block epiphysiodesis around the distal femur and proximal tibia to correct leg length differences.26 Phemister’s procedure involved removing a central block of bone from the side of the operative physis, rotating it 180 degrees and then replacing it so that the physis within the bone block was no longer aligned with the remainder of the physis. As the block healed in its new position, the physis at the operative site ultimately progressed to closure. When performed on both the medial and lateral sides of a physis, the entire physis progresses to maturity. In contrast, if the procedure is only performed on one side of the physis, the contralateral portion continues to growth, correcting the pre-existing angular deformity. This technique results in a permanent closure of the operative physis so its success in correcting deformity without over-correction is dependent on a thorough understanding of the patient’s skeletal maturity status and expected growth remaining. At the time of its publication, Phemister’s surgical technique was a huge advancement in both the treatment of leg length difference and angular extremity deformity.
Bowen at al.27 published their experience utilizing Phemister’s surgery to address significant lower extremity angular deformity. The authors emphasized that exact surgical timing is crucial for success and created a chart that included the patient’s angular deformity, gender, skeletal age (as determined by the Greulich and Pyle atlas), and data from the Green Anderson growth remaining curves to help surgeons predict the skeletal age at which a hemiepiphysiodesis should be performed.27–31 They subsequently compared outcomes among patients who had hemiepiphysiodesis performed both with and without use of their predictive chart. Use of this predictive tool to plan the surgery date resulted in appropriate angular correction without over correction at skeletal maturity. In contrast, patients in whom hemiepiphysiodesis was performed without the use of this tool had about 9 degrees of error, either in under- or over-correction of the deformity.32 McIntosh et al. reported their success with phemister-style hemiepiphysiodesis as a surgical treatment for patients with Blount disease.33 Failure of correction was defined as achieving skeletal maturity with residual mechanical axis within zone -3 or greater or residual deformity severe enough to require a corrective osteotomy. While some patients had successful angular correction with surgery, patients who were older with greater overall initial deformity or BMI ≥45 at the time of surgery were significantly more likely to fail treatment.33 Their results, again, highlight the importance of surgical timing with regards to skeletal maturity but also emphasize that other factors can impact likelihood of successful treatment with growth modulation.
The Blount Staple
In the 1930s, Venable and Stuck published their results of the effects of implanted metals and metal alloys on bone and surrounding tissue.34 They demonstrated that two metals inserted into tissue in close proximity can generate an electric current that can damage the surrounding tissue. Considering these findings, the authors recommended using metal alloys like vitallium, which is composed of cobalt, chromium, and tungsten, to minimize reaction with local tissue but maximize strength of the implant.34 Their work had significant implications for orthopaedic implant development but also served as a foundation for development of new techniques to control physeal growth.
Haas et al. hypothesized that the electric current induced by implanted metals could potentially increase physeal growth and result in a novel method for limb lengthening.35 The authors tied wire loops around all or portions of the femoral physes in an animal model and found that the loops compressed the physes, subsequently slowing growth. When the entire physis was compressed, a longitudinal discrepancy was induced, whereas angular deformity could be created by compressing only a portion of the physis.35 Further studies demonstrated that bone growth could be slowed around human physes and the effect could be reversed if the wire loop was cut and/or removed.35 Subsequent studies performed using a physeal-spanning staple also demonstrated growth retardation. However, staples that were not well fixed in bone had less of an impact on growth suppression. Authors also described multiple cases of premature ossification and noted that, even when physeal growth resumed after implant removal, it never achieved the pre-surgical growth rate.36 They hypothesized that the unpredictable growth after implant removal was related to damage to the physis at the time of implant removal. Haas et al. next investigated the use of transphyseal pins and although growth was consistently halted after pin placement, growth resumption remained inconsistent after implant removal.37
Hodgen and Frantz were the first to publish the results of using physeal-spanning staples to control growth, followed by Walter Blount in 1949.38,39 Blount recommended careful placement of multiple staples from anterior to posterior along the medial and/or lateral side of the physis to control growth (Figure 1). He also noted that growth acceleration after staple removal could be seen in some patients, making this article the first account of a phenomenon that has since been dubbed rebound deformity.39 In subsequent articles, Blount highlights that complications with this procedure occur if staples are implanted using poor technique or if patient selection is poor and emphasized that this treatment is best for younger patients, recommending permanent epiphysiodesis or hemiepiphysiodesis for patients closer to skeletal maturity.40
Figure 1. A. 13+8-year-old male with left lower extremity genu varum due to Blount disease. B and C. Intraoperative AP and lateral x-rays after placement of two lateral distal femoral and proximal tibial staples. D. Standing AP x-ray of bilateral lower extremities 13 months after staple placement, demonstrating correction of the genu varum with a neutral mechanical axis on the left side.

The staple has been widely utilized, both due to its being the oldest available implant for guided growth as well as its low cost. Raab et al. reviewed their experience with the staple for both length discrepancy and angular correction in 58 patients treated over 21 years.41 They noted excellent correction of patients with idiopathic conditions but those with “secondary deformities,” including Blount disease, had more variable results, with 68% of this cohort having only fair or poor outcomes with regards to correction.41 Implant loosening was common, occurring in 26% of surgeries with femoral staples and 35% of surgeries with proximal tibial staples. They also noted an average of about 5 degrees rebound deformity after implant removal and recommended that younger patients be allowed to slightly overcorrect to counter this response. Castañeda et al. reviewed their cumulative experience with both permanent hemiepiphysiodesis as well as staples for treatment of angular deformity.42 When patients with Blount disease were excluded from their growth modulation cohort, Castañeda at al. reported that the staple resulted in 10 degrees of correction per year from the distal femur and 7.5 degrees of correction per year from the proximal tibia.42 However, patients with Blount disease did not obtain significant correction, and the authors suggested that a permanent lateral hemiepiphysiodesis might be the better treatment in this cohort.
Percutaneous Transphyseal Screws (PETS)
In 1998, Jean-Paul Métaizeau reported his results for modulating bone growth using transphyseal screws (PETS).20 This cohort mainly consisted of patients with leg length differences in whom two screws were used to slow the growth of the entire physis to allow for equalization of leg lengths. However, he also reported that a single cancellous screw across the medial or lateral portion of the physis could be used to correct angular deformity20 (Figure 2). Métaizeau reported that 88% percent of patients began weight-bearing as tolerated within 48 hours of surgery and returned to normal activity within 15 days. The only reported growth related complications were seen in the epiphysiodesis cohort, with one patient over-correcting their leg length difference and three patients developing greater than 4 degrees of angular deformity, which Métaizeau attributed to the growth of the fibula in patients being treated for greater than 2 cm of length discrepancy.
Figure 2. A. Skeletally immature adolescent with idiopathic genu valgum from the distal femur as well as bilateral ankle valgus. B. Mechanical axis of the lower limb falls within the outer one-third of the lateral compartment bilaterally. C. Follow-up x-ray after percutaneous transepiphyseal screw placement demonstrates complete correction of the deformity with a neutral mechanical axis.

Song et al. also identified multiple technical aspects of PETS which the authors felt increased the likelihood of controlling physeal growth.22 These included (1) a more vertical screw insertion, with the goal of achieving a screw trajectory >45 degrees relative to the physis and (2) positioning screws so that at least four threads enter the epiphysis. When using screws for an epiphysiodesis to treat leg length discrepancy, the authors also recommended performing the surgery at least 1 year in advance of when the surgeon might otherwise perform an open epiphysiodesis to allow room for “lag time,” or a delay in the slowing of bone growth.22
Several small studies have analyzed using PETS to address angular deformity. Murphy et al. specifically reviewed their experiences using PETS to treat patients with genu varum due to Blount disease.43 The authors reported using 7.3 mm cannulated screws for growth modulation of tibia vara in 14 limbs (nine patients). All patients corrected their medial proximal tibial angle (MPTA), with some patients even over-correcting the MPTA to greater than 92 degrees (normal range 85-90 degrees).43 Although this study suggests that PETS can be a powerful corrective tool in the Blounts patient, the authors did not discuss any post-correction problems with the screws, such as risk of rebound deformity or risk of physeal closure after removal. Studies that have compared staples versus PETs for treating angular deformity have demonstrated similar rates of correction between the implants, with similar or reduced rates of rebound deformity after removeal.44,45 The only case of premature physeal closure occurred in a patient treated with the staple.45
Tension Band Plating
Stevens et al. was the first to report his experience using a tension band plate-screw construct (TBP) to control angular deformity46 (Figure 3). After reporting outcomes in a small group of patients with idiopathic angular deformities, he compared the results of this construct against the staple in 14 patients with deformity due to rickets.47 While patients in both groups achieved deformity correction, those treated with the staple had higher rates of implant failure and revision surgery as well as greater rates of rebound after implant removal.47 Weimann et al. compared their results with TBP and staples for growth modulation.48 They reported a 25% rate of implant-related complications among patients with pathologic physes, with similar complication rates between those treated with staples and TBP. While their results suggested possibly faster rates of correction with TBP, this difference was not statistically significant, and the rate of correction could be attributed to the younger average age of the TBP group compared with the staple group.48 TBP rapidly gained popularity and, by 2010, was reportedly the most common implant utilized for growth modulation according to a POSNA member survey.49
Figure 3. 9+2-year-old female with right lower extremity genu varum due to Blount disease. A. demonstrates her deformity prior to correction. B and C. Intraoperative images from her surgery. She underwent physeal bar resection on the medial side and placement of a tension band plate on the lateral side. A 4-hole tension band plate with non-cannulated screws was utilized. D. Postoperative alignment film demonstrating improvement alignment with a neutral mechanical axis.
Subsequent studies have confirmed that TBP is a powerful growth modulation tool with relatively low rates of peri-surgical complications. Over 900 cases of guided growth using TBP were reviewed in a multi-center study by Danino et al.50 Overall, the rate of angular correction reported was 0.77 degrees per month for the femur and 0.79 degrees per month for the tibia, with the most common complications reported being screw breakage, superficial wound dehiscence, deep infection, and limited knee range of motion postoperatively.50 As multiple patients in their study database were still skeletally immature at the time of publication, the report offered little guidance on the risk of rebound deformity or risk of premature physeal closure. A follow-up study specifically analyzing the effectiveness of TBP for managing idiopathic genu varum and valgum demonstrated significantly faster rates of correction, with an average of 0.85 degrees of correction per month around the distal femur.51 Most of the patients in this cohort were being treated for idiopathic genu valgum, and younger patients were significantly more likely to achieve neutral alignment.
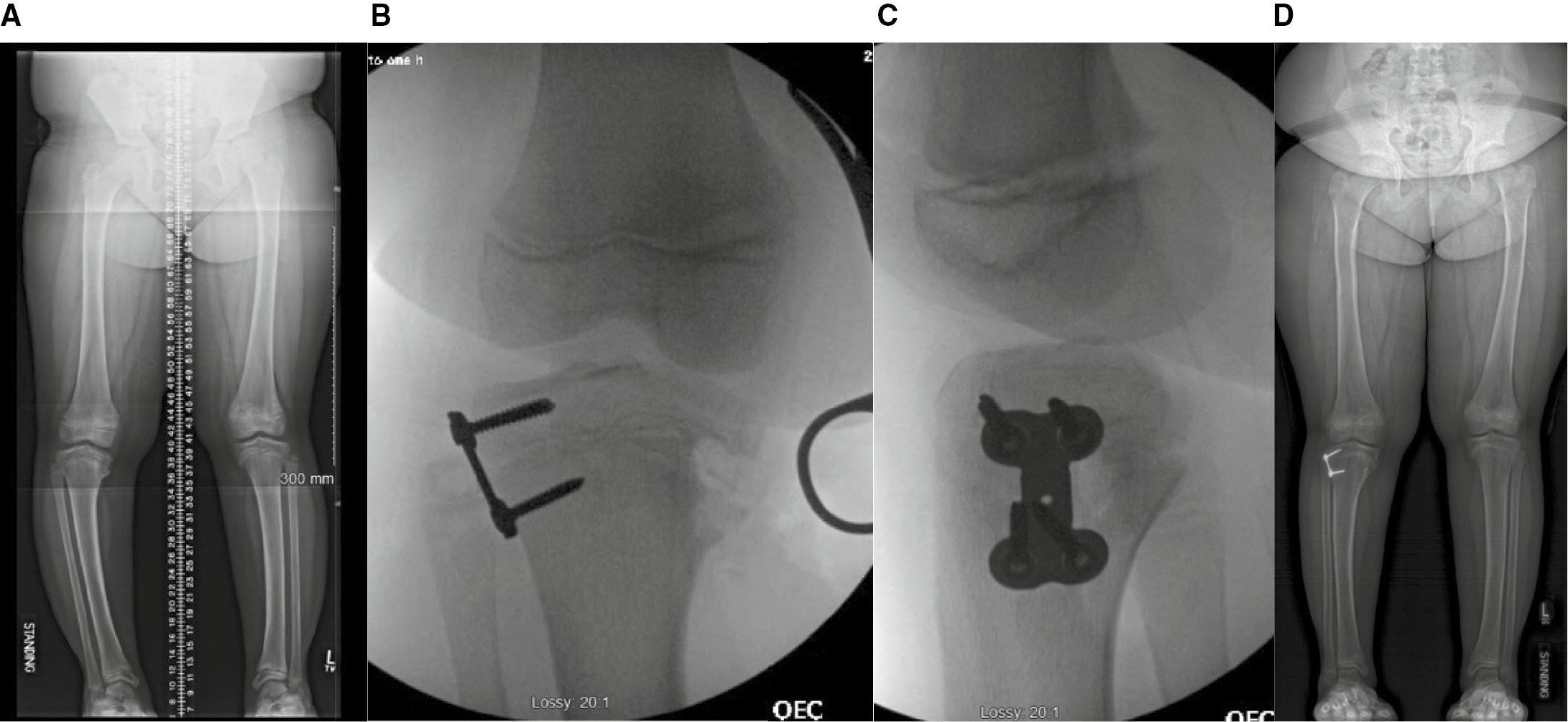
Results of growth modulation with TBP for Blount disease have been variable. In one series, TBP implant failure occurred in 50% of patients with Blount disease (Figure 4). Of the remaining Blount patients in that series, only five achieved complete correction of deformity while the remaining five obtained either no or only partial correction with this implant.52 Among POSNA members who utilize TBP, 15% reported implant failure with almost all failures occurring in titanium cannulated screws at the mid-shank of the metaphyseal screw53 (Figure 5). The average BMI among the implant failure cohort was 37, and 93% of failures occurred in patients classified as overweight, obese, or morbidly obese.53 Authors recommended increasing both the amount of fixation and utilizing non-cannulated stainless-steel screws for growth modulation in the Blounts patient. Funk et al. investigated the risk of surgical failure of growth modulation in Blount disease, comparing patients treated with either staples or TBP.54 They reported a surgical failure risk of 25% for staples and 43.3% for TBP, although this difference in surgical failure risk was not statistically significant. Among the 25 patients in their series, 24 experienced at least one perioperative complication from the growth modulation surgery.54 Similarly, Jain et al. reported an 11% mechanical failure incidence and 30% functional failure incidence in their series of patients with Blount disease treated with TBP.55 These studies suggest that while TBP offers many surgical advantages over corrective osteotomy, growth modulation is challenging in the Blount disease population regardless of implant type, with significantly greater risk of implant failure, perioperative complication, and failure of deformity correction.52–56
Figure 4. A. 5-year-old female with a history of bilateral Blount disease, status post prior bilateral proximal tibia and fibula acute osteotomies presented deformity recurrence on the right side. B. Guided growth with TBP was attempted, but she developed premature physeal arrest of the medial proximal tibial physeal and her deformity progressed, with both increased varus as well as a 2 cm limb length discrepancy. C. She ultimately underwent completion epiphysiodesis of the proximal tibial physis on the right side with right tibia and fibula osteoplasty and application of a hexapod external fixator to address her varus deformity. She also underwent right medial distal femoral guided growth with TBP to address her right distal femoral valgus. D. At skeletal maturity, she has neutral limb alignment and equalization of her leg lengths.

Figure 5. A. 6-year-old female presents with left tibia vara due to Blount disease. B. Alignment appears stable 2 months after placement of a lateral proximal tibial tension band plate using cannulated titanium screws. C. Patient presented for routine 6-month postoperative evaluation at which time it was noted that the metaphyseal screw fractured. D. Postoperative x-ray after the patient underwent revision surgery with placement 4.5 mm stainless steel screws. E. Sixteen months after revision of the tension band screws, patient presented to clinic with fracture of both the epiphyseal and diaphyseal screws.

Studies comparing TBP versus PETS have been limited. Park et al. reported a shorter duration of treatment and faster rates of correction in both the tibia and the femur with PETS compared to TBP in patients with idiopathic genu valgum.57 However, the authors did not report patient results after implant removal, so this study offers no information regarding the risk of rebound deformity or post-surgical premature physeal closure. Although Kyung et al. reported similar rates of correction between TBP and PETS, the duration between surgeries for implant placement and implant removal was shorter in the PETS group, which would suggest a faster correction with PETS.58 They did identify an increased risk of rebound deformity after TBP removal compared with PETS, and no patients experienced premature physeal closure.58 Shapiro et al. also reported faster overall rates of correction in PETS, and the surgical time for implant placement was shorter compared with TBP.59 Incision-related complications occurred in 3% of PETS and 18% of TBP patients, although this difference was not statistically significant. Hypertrophic scarring and incisional pain only occurred in the TPB group.59
Summary
Guided growth surgery has been utilized for almost a century to address angular deformity in the lower extremity. Although the technology and implants have evolved, the underlying principles are unchanged. Understanding the etiology of the deformity so that the surgeon chooses the correct operative location, precise implant placement to avoid secondary deformity, and timing the surgery with the appropriate stage of skeletal growth are critical to success. These points were all emphasized by Walter Blount and George Clark in their earliest report on the results of the staple in 1949.39 While the techniques have changed, the original reported complications with this type of surgery, including implant failure, over-correction, rebound deformity, and post-treatment premature physeal closure are the same complications we discuss with patients today.
Since its initial development, surgeons have been testing the limits and expanding the indications of growth modulation surgery. However, most of the studies evaluating guided growth are small with limited follow-up. The absence of patient follow-up through skeletal maturity greatly limits our understanding of the impact guided growth procedures have on the physis, particularly when it is used in the very skeletally immature. In addition, studies have repeatedly shown high rates of implant failure, peri-surgical complications, and failure of correction when any form of guided growth is utilized in patients who have genu varum due to Blount disease.42,52,53,55,56 While smaller incisions and a faster rehabilitation periods are tempting, the surgeon needs to consider whether the potential benefits of guided growth truly outweigh surgical risks in this population, particularly if patients have risk factors that have previously been identified as increasing the risk for failed correction.33,54
An area that has not been explored is the frequency at which guided growth creates secondary deformities in the limb. For example, guided growth has been utilized to address angular deformities due to metabolic conditions such as rickets.47 However, these metabolic conditions typically cause multi-apical diaphyseal deformity. While guided growth can be utilized to restore the mechanical axis, patients can be left with significant diaphyseal deformity, and little is known about possible risks this residual deformity poses (Figure 6). It is also possible to use guided growth to restore the mechanical axis and leave residual combined femoral and tibial valgus and varus deformity, creating a neutral limb alignment with an oblique joint line (Figure 7). There is currently no literature on the frequency with which we create this joint line obliquity and the implications this obliquity has on the long-term cartilage health for the knee are not understood. While guided growth has been utilized for decades, our current understanding of implant effectiveness and risks and the long-term effects of growth modulation on limb alignment and joint health after skeletal maturity is still limited.
Figure 6. A. 9-year-old male with bilateral genu varum due to X-linked hypophosphatemic rickets. B. TBP allowed for complete correction of the patient’s mechanical axis bilaterally, but the patient is left with significant femoral and tibial diaphyseal deformity.

Figure 7. A. Preoperative radiograph of a patient with bilateral genu varum in the setting of Blount disease. B. Bilateral TBP has restored a neutral mechanical axis bilaterally. However, closer analysis of the femurs and tibias demonstrate that this patient is left with bilateral distal femoral valgus and proximal tibial varus deformities, creating an oblique joint line in each knee.

Additional Links
- POSNA Study Guide: Hemiepiphysiodesis Technique
- American Academy of Orthopaedic Surgeons (AAOS) Orthopaedic Video Theatre: Distal Femur Guided Growth: Modified Surgical Technique With Retrograde Pin and Antegrade Screw
Disclaimer
No funding was received. The authors report no conflicts of interest related to this manuscript.
References
- McClure PK, Herzenberg JE. The natural history of lower extremity malalignment. J Pediatr Orthop. 2019;39:S14-S19.
- Frings J, Krause M, Akoto R, et al. Clinical results after combined distal femoral osteotomy in patients with patellar maltracking and recurrent dislocations. J Knee Surg. 2019;32:924-933.
- Frings J, Krause M, Akoto R, et al. Combined distal femoral osteotomy (DFO) in genu valgum leads to reliable patellar stabilization and an improvement in knee function. Knee Surg Sports Traumatol Arthrosc. 2018;26:3572-3581.
- Lin KM, Thacher RR, Apostolakos JM, et al. Implant-mediated guided growth for coronal plane angular deformity in the pediatric patient with patellofemoral instability. Arthrosc Tech. 2021;10:e913-e924.
- Nha KW, Ha Y, Oh S, et al. Surgical treatment with closing-wedge distal femoral osteotomy for recurrent patellar dislocation with genu valgum. Am J Sports Med. 2018;46:1632-1640.
- Tan SHS, Hui SJ, Doshi C, et al. The outcomes of distal femoral varus osteotomy in patellofemoral instability: a systematic review and meta-analysis. J Knee Surg. 2020;33:504-512.
- Wilson PL, Black SR, Ellis HB, et al. Distal femoral valgus and recurrent traumatic patellar instability: is an isolated varus producing distal femoral osteotomy a treatment option? J Pediatr Orthop. 2018;38:e162-e167.
- Sabharwal S. Current concepts review: blount disease. J Bone Joint Surg Am. 2009;91.
- Gordon JE, Heidenreich FP, Carpenter CJ, et al. Comprehensive treatment of late-onset tibia vara. J Bone Joint Surg Am. 2005;87-A:1561-1570.
- Iobst CA. Hexapod external fixation of tibia fractures in children. J Pediatr Orthop. 2016;36:S24-S28.
- De Bastiani G, Apley AG, Goldberg AAJ. Orthofix External Fixation in Trauma and Orthopaedics. London: Springer; 2000.
- Saran N, Rathjen KE. Guided growth for the correction of pediatric lower limb angular deformity. J Am Acad Orthop Surg. 2010;18:528-536.
- McClure PK, Kilinc E, Birch JG. Growth modulation in achondroplasia. J Pediatr Orthop. 2017;37:e384-e387.
- Gigante C, Borgo A, Corradin M. Correction of lower limb deformities in children with renal osteodystrophy by guided growth technique. J Child Orthop. 2017;11:79-84.
- Bayhan IA, Karatas AF, Rogers KJ, et al. Comparing percutaneous physeal epiphysiodesis and eight-plate epiphysiodesis for the treatment of limb length discrepancy. J Pediatr Orthop 2017;37:323-327.
- Borbas P, Agten CA, Rosskopf AB, et al. Guided growth with tension band plate or definitive epiphysiodesis for treatment of limb length discrepancy? J Orthop Surg Res. 2019;14:99.
- Gorman TM, Vanderwerff R, Pond M, et al. Mechanical axis following staple epiphysiodesis for limb-length inequality. J Bone Joint Surg Am. 2009;91:2430-2439.
- Ilharreborde B, Gaumetou E, Souchet P, et al. Efficacy and late complications of percutaneous epiphysiodesis with transphyseal screws. J Bone Joint Surg Br. 2012;94:270-275.
- Lauge-Pedersen H, Hagglund G. Eight plate should not be used for treating leg length discrepancy. J Child Orthop 2013;7:285-288.
- Metaizeau JP, Wong-Chung J, Bertrand H, et al. Percutaneous epiphysiodesis using transphyseal screws (PETS). J Pediatr Orthop. 1998;18:363-369.
- Sinha R, Weigl D, Mercado E, et al. Eight-plate epiphysiodesis: are we creating an intra-articular deformity? Bone Joint J. 2018;100-B:1112-1116.
- Song MH, Choi ES, Park MS, et al. Percutaneous epiphysiodesis using transphyseal screws in the management of leg length discrepancy: optimal operation timing and techniques to avoid complications. J Pediatr Orthop. 2015;35:89-93.
- Tolk JJ, Merchant R, Calder PR, et al. Tension-band plating for leg-length discrepancy correction. Strategies Trauma Limb Reconstr. 2022;17:19-25.
- Troy M, Shore B, Miller P, et al. A comparison of screw versus drill and curettage epiphysiodesis to correct leg-length discrepancy. J Child Orthop. 2018;12:509-514.
- Gatewood MD, Mullen BP. Experimental observations on the growth of long bones. Arch Surg (1920). 1927;15:215-221.
- Phemister DB. Operative arrestment of longitudinal growth of bones in the treatment of deformities. J Bone Joint Surg Am. 1933;15:1-15.
- Bowen JR, Leahey JL, Zhang ZH, et al. Partial epiphysiodesis at the knee to correct angular deformity. Clin Orthop Relat Res. 1985;184-190.
- Green WT, Anderson M. Experiences with epiphyseal arrest in correcting discrepancies in length of the lower extremities in infantile paralysis; a method of predicting the effect. J Bone Joint Surg Am. 1947;29:659-675.
- Green WT, Anderson M. Epiphyseal arrest for the correction of discrepancies in length of the lower extremities. J Bone Joint Surg Am. 1957;39-A:853-872; discussion, 872; passim.
- Green WT, Anderson M. Skeletal age and the control of bone growth. Instr Course Lect. 1960;17:199-217.
- Greulich WW, Pyle SI, Todd TW. Radiographic Atlas of Skeletal Development of the Hand and Wrist: Based on the Brush Foundation Study of Human Growth and Development Initiated by T. Wingate Todd. Stanford: Stanford University Press; 1950:190.
- Bowen JR, Torres RR, Forlin E. Partial epiphysiodesis to address genu varum or genu valgum. J Pediatr Orthop. 1992;12:359-364.
- McIntosh AL, Hanson CM, Rathjen KE. Treatment of adolescent tibia vara with hemiepiphysiodesis: risk factors for failure. J Bone Joint Surg Am. 2009;91:2873-2879.
- Venable CS, Stuck WG, Beach A. The effects on bone of the presence of metals; based upon electrolysis: an experimental study. Ann Surg. 1937;105:917-938.
- Haas SL. Retardation of bone growth by a wire loop. J Bone Joint Surg Am. 1945;27A:25-36.
- Haas SL. Mechanical retardation of bone growth. J Bone Joint Surg Am. 1948;30A:506-512.
- Haas SL. Restriction of bone growth by pins through the epiphyseal cartilaginous plate. J Bone Joint Surg Am. 1950;32A:338-343.
- Hodgen JT, Frantz CH. Arrest of growth of the epiphyses. Arch Surg (1920). 1946;53:664-674.
- Blount WP, Clarke GR. Control of bone growth by epiphyseal stapling; a preliminary report. J Bone Joint Surg Am. 1949;31A:464-478.
- Blount WP. A mature look at epiphyseal stapling. Clin Orthop Relat Res. 1971;77:158-163.
- Raab P, Wild A, Seller K, et al. Correction of length discrepancies and angular deformities of the leg by Blount’s epiphyseal stapling. Eur J Pediatr. 2001;160:668-674.
- Castañeda P, Urquhart B, Sullivan E, et al. Hemiepiphysiodesis for the correction of angular deformity about the knee. J Pediatr Orthop. 2008;28:188-191.
- Murphy RF, Pacult MA, Barfield WR, et al. Hemiepiphyseodesis for juvenile and adolescent tibia vara utilizing percutaneous transphyseal screws. J Pediatr Orthop. 2020;40:17-22.
- De Brauwer V, Moens P. Temporary hemiepiphysiodesis for idiopathic genua valga in adolescents: percutaneous transphyseal screws (PETS) versus stapling. J Pediatr Orthop. 2008;28:549-554.
- Black SR, Kwon MS, Cherkashin AM, et al. Lengthening in congenital femoral deficiency: a comparison of circular external fixation and a motorized intramedullary nail. J Bone Joint Surg Am. 2015;97:1432-1440.
- Stevens PM. Guided growth for angular correction: a preliminary series using a tension band plate. J Pediatr Orthop. 2007;27:253-259.
- Stevens PM, Klatt JB. Guided growth for pathological physes: radiographic improvement during realignment. J Pediatr Orthop. 2008;28:632-639.
- Wiemann JMt, Tryon C, Szalay EA. Physeal stapling versus 8-plate hemiepiphysiodesis for guided correction of angular deformity about the knee. J Pediatr Orthop. 2009;29:481-485.
- Burghardt RD, Herzenberg JE. Temporary hemiepiphysiodesis with the eight-Plate for angular deformities: mid-term results. J Orthop Sci. 2010;15:699-704.
- Danino B, Rodl R, Herzenberg JE, et al. Guided growth: preliminary results of a multinational study of 967 physes in 537 patients. J Child Orthop. 2018;12:91-96.
- Danino B, Rodl R, Herzenberg JE, et al. Growth modulation in idiopathic angular knee deformities: is it predictable? J Child Orthop. 2019;13:318-323.
- Schroerlucke S, Bertrand S, Clapp J, et al. Failure of Orthofix eight-Plate for the treatment of blount disease. J Pediatr Orthop. 2009;29:57-60.
- Burghardt RD, Specht SC, Herzenberg JE. Mechanical failures of eight-plateguided growth system for temporary hemiepiphysiodesis. J Pediatr Orthop. 2010;30:594-597.
- Funk SS, Mignemi ME, Schoenecker JG, et al. Hemiepiphysiodesis implants for late-onset Tibia Vara: a comparison of cost, surgical success, and implant failure. J Pediatr Orthop. 2016;36:29-35.
- Jain MJ, Inneh IA, Zhu H, et al. Tension Band Plate (TBP)-guided Hemiepiphysiodesis in blount disease: 10-year single-center experience with a systematic review of literature. J Pediatr Orthop. 2020;40:e138-e143.
- Oto M, Yilmaz G, Bowen JR, et al. Adolescent Blount disease in obese children treated by eight-plate hemiepiphysiodesis. Eklem Hastalik Cerrahisi. 2012;23:20-24.
- Park H, Park M, Kim SM, et al. Hemiepiphysiodesis for idiopathic genu valgum: percutaneous transphyseal screw versus tension-band plate. J Pediatr Orthop. 2018;38:325-330.
- Ko KR, Shim JS, Shin TS, et al. Factors affecting rebound phenomenon after temporary hemiepiphysiodesis and implant removal for idiopathic genu valgum in adolescent patients. J Pediatr Orthop. 2022;42:e336-e342.
- Shapiro G, Adato T, Paz S, et al. Hemiepiphysiodesis for coronal angular knee deformities: tension-band plate versus percutaneous transphyseal screw. Arch Orthop Trauma Surg. 2022;142:105-113.
